4-FA
| Summary sheet: 4-FA |
| 4-FA | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chemical Nomenclature | |||||||||||||||||||||||||||||||||
| Common names | 4-FA, 4-FMP, PAL-303, Flux | ||||||||||||||||||||||||||||||||
| Substitutive name | 4-Fluoroamphetamine, para-Fluoroamphetamine | ||||||||||||||||||||||||||||||||
| Systematic name | (RS)-1-(4-Fluorophenyl)-N-propan-2-amine | ||||||||||||||||||||||||||||||||
| Class Membership | |||||||||||||||||||||||||||||||||
| Psychoactive class | Stimulant / Entactogen | ||||||||||||||||||||||||||||||||
| Chemical class | Amphetamine | ||||||||||||||||||||||||||||||||
| Routes of Administration | |||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
| Interactions | |||||||||||||||||||||||||||||||||
| Alcohol | |||||||||||||||||||||||||||||||||
| GHB | |||||||||||||||||||||||||||||||||
| GBL | |||||||||||||||||||||||||||||||||
| Opioids | |||||||||||||||||||||||||||||||||
| Cocaine | |||||||||||||||||||||||||||||||||
| Cannabis | |||||||||||||||||||||||||||||||||
| Caffeine | |||||||||||||||||||||||||||||||||
| Ketamine | |||||||||||||||||||||||||||||||||
| Methoxetamine | |||||||||||||||||||||||||||||||||
| Psychedelics | |||||||||||||||||||||||||||||||||
| DXM | |||||||||||||||||||||||||||||||||
| PCP | |||||||||||||||||||||||||||||||||
| 25x-NBOMe | |||||||||||||||||||||||||||||||||
| 2C-T-x | |||||||||||||||||||||||||||||||||
| 5-MeO-xxT | |||||||||||||||||||||||||||||||||
| DOx | |||||||||||||||||||||||||||||||||
| Tramadol | |||||||||||||||||||||||||||||||||
| aMT | |||||||||||||||||||||||||||||||||
| MAOIs | |||||||||||||||||||||||||||||||||
4-Fluoroamphetamine (also known as 4-FA, 4-FMP, para-Fluoroamphetamine, PAL-303 and colloquially as Flux) is a novel synthetic amphetamine compound that produces a unique progressive mixture of entactogenic and stimulant effects when administered. It is part of a series of fluorinated amphetamine analog that initially included such compounds as 2-FA, 2-FMA, and 3-FA.[1]
Anecdotal reports have described the subjective effects of 4-FA as having a moderate MDMA-like entactogenic onset for the initial few hours of the experience that then gradually transitions into traditional amphetamine-type stimulation (for a total duration of around 6 to 8 hours) with residual effects that can last a few hours afterward.[citation needed]
4-FA is rarely found on the streets but was commonly sold as a grey area research chemical by online vendors along with related compounds such as 2-FMA and 3-FA.[2][1] Very little data exists about the pharmacological properties, metabolism, and toxicity of 4-FA, and it has only a brief history of human usage. Due to its strong psychostimulant effects, likely habit-forming properties as well as poorly understood toxicity profile, it is strongly recommended that one use proper harm reduction practices if choosing to use this substance.
Chemistry
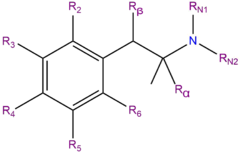
4-Fluoroamphetamine (4-FA) is a synthetic molecule of the amphetamine family. Molecules of the amphetamine class contain a phenethylamine core featuring a phenyl ring bound to an amino (NH2) group through an ethyl chain with an additional methyl substitution at Rα. Amphetamines are alpha-methylated phenethylamines. 4-fluoroamphetamine contains a fluorine atom at R4 of its phenyl ring and is a fluorinated analogue of amphetamine.
Pharmacology
4-Fluoroamphetamine acts as a releasing agent and reuptake inhibitor of dopamine, serotonin, and norepinephrine producing stimulating amphetamine-like effects at lower doses and euphoric, entactogenic effects similar to MDMA at dosages above 100mg. The mechanism of action of 4-FA effectively boosts the levels of the norepinephrine, dopamine, and serotonin neurotransmitters in higher doses in the brain by binding to and partially blocking the transporter proteins that normally remove those monoamines from the synaptic cleft. This allows dopamine, norepinephrine, and serotonin to accumulate within the brain, resulting in stimulating, euphoric and entactogenic effects.[3][2]
Subjective effects
It is commonly reported that the first three to four hours of 4-FA present distinct entactogenic effects that have been reported as feeling somewhat similar to MDMA although not quite as powerful. This is thought to correlate with the duration in which it is promoting the release of serotonin (in addition to dopamine and norepinephrine). After this first phase of the experience, the effect then shifts towards something which feels like classic amphetamine stimulation which can persist for an extended period.[citation needed]
Do not use 4-FA if you have a history of heart-related issues or experience a severe headache after its use. We have been made aware of a report released by Trimbos-instituut[4] and Nationaal Vergiftigingen Informatie Centrum[5] (NVIC), describing incidents of strokes after an increased use of 4-FA. In addition to the common amphetamine-like effects (agitation, anxiety, tachycardia, hypertension, chest pain et al.), serious cardio- and cerebrovascular complications have been reported, including rhythm (sinus arrhythmia, ventricular extrasystoles (bigeminy), conduction disturbances) and acute cardiac failure. Although a causal relationship has not been confirmed, when presented with a severe headache and lateralization after 4-FA usage, a medical evaluation at an emergency department should be conducted immediately. [6]
Disclaimer: The effects listed below cite the Subjective Effect Index (SEI), an open research literature based on anecdotal user reports and the personal analyses of PsychonautWiki contributors. As a result, they should be viewed with a healthy degree of skepticism.
It is also worth noting that these effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects. Likewise, adverse effects become increasingly likely with higher doses and may include addiction, severe injury, or death ☠.
Physical effects 
-
- Stimulation - The first few hours of the 4-FA experience are commonly reported to have overtones of the type of sedation that is associated with the serotonin-releasing properties of entactogens like MDMA. After these entactogenic effects fade and the stimulating effects become predominant. The stimulation which 4-FA produces throughout the entirety of the experience be described as being slightly weaker in intensity to amphetamine and less forceful than traditional dopaminergic stimulants such as cocaine.
- Spontaneous bodily sensations - The "body high" of 4-FA can be characterized as a moderate to extreme euphoric tingling sensation that encompasses the entire body. It is capable of becoming overwhelmingly pleasurable at higher doses. This sensation maintains a consistent presence that steadily rises with the onset and hits its limit once the peak has been reached.
- Physical euphoria - This effect is extremely intense when compared to its physical stimulation.
- Tactile enhancement
- Bodily control enhancement
- Stamina enhancement
- Temperature regulation suppression
- Abnormal heartbeat
- Increased heart rate
- Increased blood pressure
- Vibrating vision - At high doses, a person's eyeballs may begin to spontaneously wiggle back and forth in a rapid motion, causing the vision to become blurry and temporarily out of focus. This is a condition known as nystagmus and is considered to be harmless in most cases.
- Headaches
- Muscle contractions
- Appetite suppression
- Dehydration
- Dry mouth
- Increased perspiration
- Pupil dilation
- Orgasm suppression
- Temporary erectile dysfunction
- Difficulty urinating
- Teeth grinding
- Seizure - This is a rare effect but is thought to be able to occur in those predisposed to them, especially when taking heavier-than-recommended doses or redosing while in physically taxing conditions such as being dehydrated, fatigued, undernourished, or overheated.[citation needed]
Visual effects 
-
Enhancements
Auditory effects 
Cognitive effects 
-
- Anxiety suppression
- Disinhibition
- Empathy, affection, and sociability enhancement - In comparison to other substances, this effect can be described as identical to the effects produced by MDMA, but with less intensity. Like euphoria, it is most present at dosages above 100mg.
- Cognitive euphoria - This effect can be described in its manifestation as a series of euphoric waves that recede and reappear randomly throughout the experience. It is most present at dosages above 100mg.
- Thought acceleration
- Focus enhancement
- Novelty enhancement
- Immersion enhancement
- Motivation enhancement
- Increased music appreciation
- Increased sense of humor
- Compulsive redosing
- Increased libido
- Delirium & Confusion - This effect typically only occurs with overly high doses, and is associated with temperature dysregulation and overheating, particularly when 4-FA is taken in crowded, physically strenuous environments that leaves the user unable to cool off, rest, or rehydrate adequately.
- Ego inflation
- Time distortion - This can be described as the experience of time speeding up and passing much quicker than it usually would when sober.
- Wakefulness
Transpersonal effects 
-
- Unity and interconnectedness - Experiences of unity, oneness, and interconnectedness are sometimes reported with 4-FA. This component most consistently manifests itself at higher doses during the empathogenic phase, within large crowds at raves and musical events in the form of "becoming one with the crowd." Music is said to intensify this effect as well consistently.
After effects 
-
The effects which occur during the offset of a stimulant experience generally feel negative and uncomfortable in comparison to the effects which occurred during its peak. This is often referred to as a "comedown" and occurs because of neurotransmitter depletion. Its effects commonly include:
- Anxiety
- Brain zaps
- Headaches
- Cognitive fatigue
- Depression
- Sleep paralysis - Some users report a higher incidence of experiencing sleep paralysis after consuming 4-FA, usually only at higher doses.
- Irritability
- Motivation suppression
- Thought deceleration
- Thought disorganization
- Suicidal ideation - This effect is typically only reported when 4-FA is misused by either taking it too frequently, with excessive doses, and/or compulsively.
- Wakefulness
Experience reports
Anecdotal reports which describe the effects of this compound within our experience index include:
- Experience:120mg/170mg 4-FA - Substance Overview
- Experience:500mg 4-FA + 150mg 5-HTP - Irresponsible & unexpected psychedelic
Additional experience reports can be found here:
Toxicity and harm potential
The toxicity and long-term effects of recreational 4-FA use have only scarcely been studied because 4-FA has very little history of human usage. Anecdotal evidence, as well as several case reports, suggest there is a small to moderate individual health risk associated with the use of 4-FA. Among these, it appears there is an especially a high risk for acute cardiovascular toxicity. Some people in the Netherlands, only using a moderate dose, have died due to cardiac arrest or suffered severe brain damage due to a stroke. The mechanism of this acute toxicity is not yet known, but it seems as though the stroke starts off as an intense headache or even a migraine attack, that slowly worsens.[7]
The LD50 (mouse; i.p.) of 4-FA is 46 mg/kg.[8] 4-FA does not cause long-lasting depletion of brain serotonin unlike MDMA or 4-FA's analogs 4-CA and 4-BA.
It is also worth noting that 4-FA is particularly caustic in comparison to other compounds and can, therefore, cause chemical burns within the nasal passage and throat if it is insufflated. This method of administration is discouraged.
It is strongly recommended that one use harm reduction practices when using this substance.
Tolerance and addiction potential
As with other stimulants, the chronic use of 4-FA can be considered moderately addictive with a high potential for abuse and is capable of causing psychological dependence among certain users. When addiction has developed, cravings and withdrawal effects may occur if a person suddenly stops their usage.
Tolerance to many of the effects of 4-FA develops with prolonged and repeated use. This results in users having to administer increasingly large doses to achieve the same effects. After that, it takes about 3 - 7 days for the tolerance to be reduced to half and 1 - 2 weeks to be back at baseline (in the absence of further consumption). This is how long it takes to reduce the tolerance for the stimulating effects. Tolerance for the entactogenic effects may take a longer period to reduce. 4-FA presents cross-tolerance with all dopaminergic stimulants, meaning that after the consumption of 4-FA all stimulants will have a reduced effect.
Psychosis
4-FA, like other stimulants, can result in a stimulant psychosis that may present with a variety of symptoms (e.g., paranoia, hallucinations, or delusions).[9][10] A review on treatment for amphetamine, dextroamphetamine, and methamphetamine abuse-induced psychosis states that about 5–15% of users fail to recover completely.[10][11] The same review asserts that, based upon at least one trial, antipsychotic medications effectively resolve the symptoms of acute amphetamine psychosis.[10]
Dangerous interactions
Warning: Many psychoactive substances that are reasonably safe to use on their own can suddenly become dangerous and even life-threatening when combined with certain other substances. The following list provides some known dangerous interactions (although it is not guaranteed to include all of them).
Always conduct independent research (e.g. Google, DuckDuckGo, PubMed) to ensure that a combination of two or more substances is safe to consume. Some of the listed interactions have been sourced from TripSit.
- Alcohol - Drinking alcohol on stimulants is considered risky because it reduces the sedative effects of the alcohol that the body uses to gauge drunkenness. This often leads to excessive drinking with greatly reduced inhibitions, increasing the risk of liver damage and increased dehydration. The effects of stimulants will also allow one to drink past a point where they might normally pass out, increasing the risk. If you do decide to do this then you should set a limit of how much you will drink each hour and stick to it, bearing in mind that you will feel the alcohol and the stimulant less.
- GHB/GBL - Stimulants increase respiration rate allowing a higher dose of sedatives. If the stimulant wears off first then the depressant effects of the GHB/GBL may overcome the user and cause respiratory arrest.
- Opioids - Stimulants increase respiration rate allowing a higher dose of opiates. If the stimulant wears off first then the opiate may overcome the patient and cause respiratory arrest.
- Cocaine - The rewarding effects of cocaine are mediated by DAT inhibition, and an increase of exocytosis of dopamine through the cell membrane. Amphetamine reverses the direction of DAT and the direction vesicular transports within the cell by a pH mediated mechanism of displacement, thus excludes the regular mechanism of dopamine release through means of exocytosis because the effects Na+/K+ ATPase are inhibited. You will find cardiac effects with the combination of cocaine and amphetamine due to a SERT mediated mechanism from the subsequent activation of 5-HT2B, which is an effect of serotonin-related valvulopathy. Amphetamines generally cause hypertension in models of abuse, and this combination can increase the chances of syncope due to turbulent blood flow during valve operation. The rewarding mechanisms of cocaine are reversed by administration of amphetamine.[12][13]
- Cannabis - Stimulants increase anxiety levels and the risk of thought loops and paranoia which can lead to negative experiences.
- Caffeine - This combination of stimulants is generally considered unnecessary and may increase strain on the heart, as well as potentially causing anxiety and physical discomfort.
- Tramadol - Tramadol and stimulants both increase the risk of seizures.
- DXM - Both substances raise heart rate, in extreme cases, panic attacks caused by these substances have led to more serious heart issues.
- Ketamine - Combining amphetamine and ketamine may result in psychoses that resemble schizophrenia, but not worse than the psychoses produced by either substance alone, but this is debatable. This is due to amphetamines ability to attenuated the disruption of working memory caused by ketamine. Amphetamine alone may result in grandiosity, paranoia, or somatic delusions with little to no effect on negative symptoms. Ketamine, however, will result in thought disorders, disruption of executive functioning, and delusions due to a modification of conception. These mechanisms are due to an increase of dopaminergic activity in the mesolimbic pathway caused by amphetamine due to its pharmacology effecting dopamine, and due to a disruption of dopaminergic functioning in the mesocortical pathways via NMDA antagonism effects of ketamine. Combining the two, you may expect mainly thought disorder along with positive symptoms.[14]
- PCP - Increases risk of tachycardia, hypertension, and manic states.
- Methoxetamine - Increases risk of tachycardia, hypertension, and manic states.
- Psychedelics (e.g. LSD, mescaline, psilocybin) - Increases risk of anxiety, paranoia, and thought loops.
- 25x-NBOMe - Amphetamines and NBOMes both provide considerable stimulation that when combined they can result in tachycardia, hypertension, vasoconstriction and, in extreme cases, heart failure. The anxiogenic and focusing effects of stimulants are also not good in combination with psychedelics as they can lead to unpleasant thought loops. NBOMes are known to cause seizures and stimulants can increase this risk.
- 2C-T-x - Suspected of mild MAOI properties. May increase the risk of hypertensive crisis.
- 5-MeO-xxT - Suspected of mild MAOI properties. May increase the risk of hypertensive crisis.
- DOx
- aMT - aMT has MAOI properties which may interact unfavorably with amphetamines.
- MAOIs - MAO-B inhibitors can increase the potency and duration of phenethylamines unpredictably. MAO-A inhibitors with amphetamine can lead to hypertensive crises.
Legal status
- Austria: 4-FA is illegal to possess, produce and sell under the NPSG (Neue-Psychoaktive-Substanzen-Gesetz Österreich).[citation needed]
- Brazil: 4-FA is illegal to possess, produce and sell as it is listed on Portaria SVS/MS nº 344.[15]
- Canada: 4-FA would fall under Schedule I as it is considered an analog of amphetamine.[16]
- France: 4-FA is scheduled as a "stupéfiant", i.e. a recognized drug of abuse. It is illegal to possess, buy, sell or manufacture.[17]
- Germany: 4-FA is controlled under Anlage I BtMG (Narcotics Act, Schedule I)[18] as of July 26, 2012.[19] It is illegal to manufacture, possess, import, export, buy, sell, procure or dispense it without a license.[20]
- Hungary: In January 2012, 4-FA became controlled in Hungary.[citation needed]
- Israel: In December 2007, 4-FA was added to Israel's list of controlled substances, making it illegal to buy, sell, or possess.[citation needed]
- The Netherlands: 4-FA is a Schedule 1 drug in the Netherlands as of May 25th 2017. [21]
- New Zealand: 4-FA is an amphetamine analogue, so is a Schedule 3 controlled substance in New Zealand.[22]
- Poland: 4-FA is controlled in Poland.[citation needed]
- Slovak Republic: Beginning March 1, 2011, 4-Fluoroamphetamine is controlled in the Slovak Republic.[citation needed]
- Switzerland: 4-FA is a controlled substance specifically named under Verzeichnis D.[23]
- Turkey: 4-FA is a classed as drug and is illegal to possess, produce, supply, or import.[24] [25]
- United Kingdom: 4-FA is considered a Class A drug as a result of the amphetamine analogue clause of the Misuse of Drugs Act 1971.[26]
- United States: 4-FA is not scheduled on a federal level in the United States.[citation needed]
- Arizona: 4-FA was added to the "Dangerous Drug" list in April 2014.[citation needed]
- Louisiana: 4-FA is currently listed as a Schedule I drug as of June 2013.[citation needed]
- Virginia: 4-FA is classified as a Schedule I drug.[citation needed]
See also
External links
Discussion
References
- ↑ 1.0 1.1 Rösner, P., Quednow, B., Girreser, U., Junge, T. (March 2005). "Isomeric Fluoro-methoxy-phenylalkylamines: a new series of controlled-substance analogues (designer drugs)". Forensic Science International. 148 (2–3): 143–156. doi:10.1016/j.forsciint.2004.05.003. ISSN 0379-0738.
- ↑ 2.0 2.1 Nagai, F., Nonaka, R., Satoh Hisashi Kamimura, K. (22 March 2007). "The effects of non-medically used psychoactive drugs on monoamine neurotransmission in rat brain". European Journal of Pharmacology. 559 (2–3): 132–137. doi:10.1016/j.ejphar.2006.11.075. ISSN 0014-2999.
- ↑ Fuller, R. W., Baker, J. C., Perry, K. W., Molloy, B. B. (1 October 1975). "Comparison of 4-chloro-, 4-bromo- and 4-fluoroamphetamine in rats: Drug levels in brain and effects on brain serotonin metabolism". Neuropharmacology. 14 (10): 739–746. doi:10.1016/0028-3908(75)90099-4. ISSN 0028-3908.
- ↑ Voor mentale gezondheid
- ↑ Vergiftigingen.info - Home
- ↑ https://psychonautwiki.org/wiki/File:Behandeling-4-fa-intoxicatie.pdf
- ↑ https://psychonautwiki.org/wiki/File:4-FA%20risicobeoordeling%20(2016).pdf
- ↑ Costa, E., Garattini, S. (June 1971). Domino, E. F., ed. "Amphetamines and related compounds". Electroencephalography and Clinical Neurophysiology. 30 (6): 579. doi:10.1016/0013-4694(71)90160-X. ISSN 0013-4694.
- ↑ National Institute on Drug Abuse, Emerging Trends
- ↑ 10.0 10.1 10.2 Shoptaw, S. J., Kao, U., Ling, W. (21 January 2009). Cochrane Drugs and Alcohol Group, ed. "Treatment for amphetamine psychosis". Cochrane Database of Systematic Reviews. doi:10.1002/14651858.CD003026.pub3. ISSN 1465-1858.
- ↑ Hofmann, F. G. (1983). A handbook on drug and alcohol abuse: the biomedical aspects (2nd ed ed.). Oxford University Press. ISBN 9780195030563.
- ↑ Greenwald, M. K., Lundahl, L. H., Steinmiller, C. L. (December 2010). "Sustained Release d-Amphetamine Reduces Cocaine but not 'Speedball'-Seeking in Buprenorphine-Maintained Volunteers: A Test of Dual-Agonist Pharmacotherapy for Cocaine/Heroin Polydrug Abusers". Neuropsychopharmacology. 35 (13): 2624–2637. doi:10.1038/npp.2010.175. ISSN 0893-133X.
- ↑ Siciliano, C. A., Saha, K., Calipari, E. S., Fordahl, S. C., Chen, R., Khoshbouei, H., Jones, S. R. (10 January 2018). "Amphetamine Reverses Escalated Cocaine Intake via Restoration of Dopamine Transporter Conformation". The Journal of Neuroscience. 38 (2): 484–497. doi:10.1523/JNEUROSCI.2604-17.2017. ISSN 0270-6474.
- ↑ Krystal, J. H., Perry, E. B., Gueorguieva, R., Belger, A., Madonick, S. H., Abi-Dargham, A., Cooper, T. B., MacDougall, L., Abi-Saab, W., D’Souza, D. C. (1 September 2005). "Comparative and Interactive Human Psychopharmacologic Effects of Ketamine and Amphetamine: Implications for Glutamatergic and Dopaminergic Model Psychoses and Cognitive Function". Archives of General Psychiatry. 62 (9): 985. doi:10.1001/archpsyc.62.9.985. ISSN 0003-990X.
- ↑ http://portal.anvisa.gov.br/documents/10181/3115436/%281%29RDC_130_2016_.pdf/fc7ea407-3ff5-4fc1-bcfe-2f37504d28b7
- ↑ Branch, L. S. (2022), Consolidated federal laws of Canada, Controlled Drugs and Substances Act
- ↑ Arrêté du 22 février 1990 fixant la liste des substances classées comme stupéfiants
- ↑ "Anlage I BtMG" (in German). Bundesministerium der Justiz und für Verbraucherschutz. Retrieved December 18, 2019.
- ↑ "Sechsundzwanzigste Verordnung zur Änderung betäubungsmittelrechtlicher Vorschriften" (in German). Bundesanzeiger Verlag. Retrieved December 18, 2019.
- ↑ "§ 29 BtMG" (in German). Bundesministerium der Justiz und für Verbraucherschutz. Retrieved December 18, 2019.
- ↑ https://www.rijksoverheid.nl/actueel/nieuws/2017/05/24/verbod-op-drug-4-fluoramfetamine-gaat-vrijdag-25-mei-in
- ↑ Misuse of Drugs Act 1975 No 116 (as at 01 July 2022), Public Act – New Zealand Legislation
- ↑ "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien" (in German). Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved January 1, 2020.
- ↑ Başbakanlık Mevzuatı Geliştirme ve Yayın Genel Müdürlüğü
- ↑ https://resmigazete.gov.tr/eskiler/2014/01/20140125-3-1.pdf
- ↑ Misuse of Drugs Act 1971