2C-I
| Summary sheet: 2C-I |
| 2C-I | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chemical Nomenclature | |||||||||||||||||||||||||||||||||
| Common names | 2C-I | ||||||||||||||||||||||||||||||||
| Substitutive name | 2,5-Dimethoxy-4-iodophenethylamine | ||||||||||||||||||||||||||||||||
| Systematic name | 2-(4-Iodo-2,5-dimethoxyphenyl)ethan-1-amine | ||||||||||||||||||||||||||||||||
| Class Membership | |||||||||||||||||||||||||||||||||
| Psychoactive class | Psychedelic | ||||||||||||||||||||||||||||||||
| Chemical class | Phenethylamine | ||||||||||||||||||||||||||||||||
| Routes of Administration | |||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
| Interactions | |||||||||||||||||||||||||||||||||
| Cannabis | |||||||||||||||||||||||||||||||||
| Stimulants | |||||||||||||||||||||||||||||||||
| Tramadol | |||||||||||||||||||||||||||||||||
| Lithium | |||||||||||||||||||||||||||||||||
2,5-Dimethoxy-4-iodophenethylamine (also known as 2C-I and "Smiles") is a psychedelic substance of the phenethylamine class. It is a member of the 2C-x family of psychedelic phenethylamines, all of which were derived from the systematic modification of the mescaline molecule.[1]
2C-I was first synthesized and investigated for human activity by Alexander Shulgin in 1976[2] and later described in his 1991 book PiHKAL ("Phenethylamines I Have Known and Loved"). Its PiHKAL entry notes it for being "very colorful and active" but without "feelings of insight, revelation, or progress toward the true meaning of the universe."[1] It first briefly gained some popularity in the late 1990s and early 2000s, where it was sold in several smart shops as a replacement for 2C-B following its scheduling in 1995.[3] It also has a history of being sold online as a research chemical before becoming scheduled.
2C-I is typically used recreationally for its psychedelic as well as moderate stimulating and entactogenic effects. It has been explored as a potential stimulant nootropic in the 1-8mg dosage range.[citation needed]
Little data exists about the pharmacological properties, metabolism, and toxicity of 2C-I, and it has a limited history of human use. 2C-I is sometimes confused for the analog 25I-NBOMe (an abbreviated form of 2C-I-NBOMe). It is highly advised to use harm reduction practices if using this substance.
Chemistry
This chemistry section is incomplete. You can help by adding to it. |
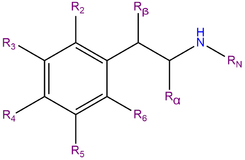
2C-I, or 2,5-dimethoxy-4-iodophenethylamine, is a substituted phenethylamine comprising a phenyl ring bound to an amino (NH2) group through an ethyl chain. 2C-I contains methoxy functional groups CH3O- attached to carbons R2 and R5 as well as an iodine atom attached to carbon R4 of the phenyl ring. 2C-I belongs to the 2C family of phenethylamines which contain methoxy groups on the 2 and 5 positions of the benzene ring.
Pharmacology
 |
This pharmacology section is incomplete. You can help by adding to it. |
2C-I is a psychedelic phenethylamine of the 2C-x family. 2C-I is taken orally in tablet or capsule forms or snorted in its powder form. It has also been found impregnated on small squares of blotter paper for oral administration, which is a technique often seen for the distribution and abuse of LSD. 2C-I primarily causes stimulant and hallucinogenic effects, including increased visual, auditory, and tactile sensations. Moderate doses can cause full hallucinations and can also produce a euphoric high.
2C-I's psychedelic effects are believed to come from its efficacy at the 5-HT2A receptor as a partial agonist. However, the role of these interactions and how they result in the psychedelic experience remains the subject of ongoing scientific investigation.
Subjective effects
Disclaimer: The effects listed below cite the Subjective Effect Index (SEI), an open research literature based on anecdotal user reports and the personal analyses of PsychonautWiki contributors. As a result, they should be viewed with a healthy degree of skepticism.
It is also worth noting that these effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects. Likewise, adverse effects become increasingly likely with higher doses and may include addiction, severe injury, or death ☠.
Physical effects 
-
- Stimulation - In terms of its effects on the physical energy levels of the user, 2C-I is usually considered to be very energetic and stimulating in a fashion that is quite comparable to MDMA, although it is 'encouraged' instead of 'forced'.
- Spontaneous physical sensations - The "body high" of 2C-I is manifested as one of the most proportionally intense in comparison to almost all of the classical psychedelics. The sensation itself can be described as an intense and slightly uncomfortable energetic pins and needles sensation that constantly encompasses a person’s entire body. It is usually felt over every square inch of the skin but occasionally manifests itself in the form of a continuously shifting, tingling sensation that travels up and down the body in spontaneous waves. Alongside this, many users commonly report that the "body high" can be particularly uncomfortable and sometimes accompanied by dysphoric aches and urges to shift the position of one's body and prolonged tensing of unusual combinations of muscle groups.
- Physical euphoria - Feelings of frequent but unpredictable rushes of warm physical euphoria are extremely common. These can move from the top of the head downwards before enveloping one's whole body.
- Tactile enhancement
- Bodily control enhancement
- Stamina enhancement
- Increased heart rate
- Temperature regulation suppression
- Muscle contractions
- Muscle cramps
- Muscle spasms
- Appetite suppression
- Dehydration
- Frequent urination
- Nausea - Mild to extreme nausea is consistently reported when consumed in moderate to high doses and either passes once the user has vomited or gradually fades by itself as the peak sets in.
- Pupil dilation
Visual effects 
-
Enhancements
Distortions
- Drifting (melting, flowing, breathing and morphing) - In comparison to other psychedelics, this effect can be described as simplistic and bland in detail, slow and smooth but sometimes jittery in motion, static in appearance and unrealistic/cartoon-like in style.
- Colour shifting
- Environmental cubism - This effect is unique in that is it usually manifests simultaneously alongside or directly as a result of external geometry
- Environmental patterning
- Scenery slicing
- Symmetrical texture repetition
- Tracers
Geometry
The visual geometry that is present throughout this trip can be described as more similar in appearance to that of LSD or 2C-B than that of 2C-E, psilocin, or ayahuasca although it is much blander and less detailed. They can be comprehensively described as unstructured in their organization, algorithmic in geometric style, intricate in complexity, large in size, fast and smooth in motion, colorful in scheme, glossy in color, both soft and sharp in their edges as well as equally rounded and angular in their corners. They seem high in algorithmic visuals such as fractals and at higher doses are significantly more likely to result in states of level 8A visual geometry over level 8B.
Hallucinatory states
Like LSD, while 2C-I is capable of producing a full range of low and high level hallucinatory states, this is extremely rare and inconsistent at higher levels but common at lower ones.
- Transformations
- Internal hallucination (autonomous entities; settings, sceneries, and landscapes; perspective hallucinations and scenarios and plots) - Although 2C-I is technically capable of producing hallucinatory states in a fashion that is on par with psilocin or DMT in its vividness and intensity, in comparison, these effects are extremely rare and inconsistent. Whilst traditional psychedelics such as LSA, ayahuasca and mescaline will induce internal hallucinations near consistently at level 5 geometry and above, 2C-I hallucinations will not extend beyond imagery and higher doses will for most simply go straight into level 8A visual geometry. This lack of consistently induced hallucinatory breakthroughs means that, for most, 2C-I is not as deep of an experience as certain other psychedelics.
- External hallucination (autonomous entities; settings, sceneries, and landscapes; perspective hallucinations and scenarios and plots)
Cognitive effects 
-
The head space of 2C-I is described by many as one which is relatively normal in its thought processes even at moderate to high doses. It is often said to lack insight when compared to that of 2C-E, psilocin and LSD.
- Empathy, affection, and sociability enhancement - This component is consistently manifested only in the context of social settings in which one is within the company of others. These feelings of sociability, affection and empathy are a little weaker and less sharp than those found on substances such as MDMA and 2C-B but still prove strong enough to provide long-lasting therapeutic effects.
- Analysis enhancement
- Emotion enhancement or Emotion suppression
- Immersion enhancement
- Increased libido
- Increased music appreciation
- Memory suppression
- Novelty enhancement
- Time distortion
- Wakefulness
Auditory effects 
Multi-sensory effects 
-
- Synaesthesia - In its fullest manifestation, this is a very rare and non-reproducible effect. Increasing the dosage can increase the likelihood of this occurring, but seems to only be a prominent part of the experience among those who are already predisposed to synaesthetic states.
Transpersonal effects 
-
- Unity and interconnectedness In contrast to some other psychedelics, this effect does not readily occur except at very high doses.
Experience reports
Anecdotal reports which describe the effects of this compound within our experience index include:
Additional experience reports can be found here:
Toxicity and harm potential
 |
This toxicity and harm potential section is a stub. As a result, it may contain incomplete or even dangerously wrong information! You can help by expanding upon or correcting it. |
The toxicity and long-term health effects of recreational 2C-I use do not seem to have been studied in any scientific context and the exact toxic dose is unknown. This is because 2C-I is a research chemical with very little history of human usage.
Anecdotal reports from those who have tried 2C-I suggests that there are no negative health effects attributed to simply trying the substance by itself at low to moderate doses and using it very sparingly (but nothing can be completely guaranteed). Independent research should always be done to ensure that a combination of two or more substances is safe before consumption.
It is strongly recommended that one use harm reduction practices, such as volumetric dosing, when using this substance so as to ensure the accurate administration of the intended dose.
Tolerance and addiction potential
Although no formal studies have been conducted, it is not unreasonable to assume that as is the case with psychedelics in general, 2C-I is not habit-forming and that the desire to use it can actually decrease with use.
Tolerance to the effects of 2C-I is built almost immediately after ingestion. After that, it takes about 3-5 days for the tolerance to be reduced to half and 7-10 days to be back at baseline (in the absence of further consumption). 2C-I presents cross-tolerance with all psychedelics, meaning that after the consumption of 2C-I all psychedelics will have a reduced effect.
Dangerous interactions
Warning: Many psychoactive substances that are reasonably safe to use on their own can suddenly become dangerous and even life-threatening when combined with certain other substances. The following list provides some known dangerous interactions (although it is not guaranteed to include all of them).
Always conduct independent research (e.g. Google, DuckDuckGo, PubMed) to ensure that a combination of two or more substances is safe to consume. Some of the listed interactions have been sourced from TripSit.
- Lithium - Lithium is commonly prescribed for the treatment of bipolar disorder. There is a large body of anecdotal evidence that suggests taking it with psychedelics significantly increases the risk of psychosis and seizures. As a result, this combination is strictly discouraged.
- Cannabis - Cannabis may have an unexpectedly strong and unpredictable synergy with the effects of 2C-I. Caution is advised with this combination as it can significantly increase the risk of adverse psychological reactions like anxiety, paranoia, panic attacks, and psychosis. Users are advised to start off with only a fraction of their normal cannabis dose and take long breaks between hits to avoid unintentional overdose.
- Stimulants - Stimulants like amphetamine, cocaine or methylphenidate affect many parts of the brain and alter dopaminergic function. This combination can increase the risk of anxiety, paranoia, panic attacks, and thought loops. This interaction may also result in an elevated risk of mania and psychosis.[citation needed]
- Tramadol - Tramadol is well-documented to lower the seizure threshold[4] and psychedelics may act to trigger seizures in susceptible individuals.[citation needed]
Legal status
In November 2003, the European Council decided that 2C-I shall be subjected by the Member States to control measures and criminal penalties within three months.[5]
- Austria: 2C-I is illegal to possess, produce and sell under the SMG. (Suchtmittelgesetz Österreich)[citation needed]
- Brazil: Possession, production and sale is illegal as it is listed on Portaria SVS/MS nº 344.[6]
- Canada: 2C-I is a schedule III controlled substance in Canada.[7]
- Denmark: 2C-I is a controlled substance in Denmark.[8]
- Germany: 2C-I is controlled under Anlage I BtMG[9] (Narcotics Act, Schedule I) as of October 10, 1999.[10] It is illegal to manufacture, possess, import, export, buy, sell, procure or dispense it without a license.[11]
- Greece: 2C-I is a controlled substance in Greece.[8]
- Ireland: 2C-I is a controlled substance in Ireland.[8]
- Italy: 2C-I is a controlled substance in Italy.[8]
- Latvia: 2C-I is a Schedule I controlled substance Latvia.[12]
- The Netherlands: 2C-I is a controlled substance in the Netherlands.[8]
- Poland: 2C-I is a controlled substance in Poland.[8]
- Sweden: Sveriges riksdag added 2C-I to Schedule I ("substances, plant materials and fungi which normally do not have medical use") as a narcotic in Sweden as of March 16, 2004.[13]
- Switzerland: 2C-I is a controlled substance specifically named under Verzeichnis D.[14]
- Turkey: 2C-I is classed as a drug and is illegal to possess, produce, supply, or import.[15]
- United Kingdom: 2C-I is a Class A drug in the United Kingdom as a result of the phenethylamine catch-all clause.[16]
- United States: As of July 9, 2012, 2C-I is a Schedule I substance in the U.S. under the Synthetic Drug Abuse Prevention Act of 2012, making possession, distribution and manufacture illegal.[8]
See also
External links
Community
References
- ↑ 1.0 1.1 Alexander Shulgin; Ann Shulgin (1991). "#33. 2C-I". PiHKAL: A Chemical Love Story. United States: Transform Press. ISBN 0963009605. OCLC 1166889264.
- ↑ Alexander Shulgin. Pharmacology Notes I (The Shulgin Lab Books) (PDF). Erowid. p. 177.
- ↑ Eric Johnson (March 19, 2011). Ellen Wulfhorst, ed. "Synthetic drug, subject of proposed bans, kill teen". Reuters. Retrieved October 11, 2020.
- ↑ Talaie, H.; Panahandeh, R.; Fayaznouri, M. R.; Asadi, Z.; Abdollahi, M. (2009). "Dose-independent occurrence of seizure with tramadol". Journal of Medical Toxicology. 5 (2): 63–67. doi:10.1007/BF03161089. ISSN 1556-9039.
- ↑ "COUNCIL DECISION 2003/847/JHA". Official Journal of the European Union. Office for Official Publications of the European Communites (published December 6, 2003). November 27, 2003. pp. 64–65. OCLC 52224955. L 321.
- ↑ "RESOLUÇÃO DA DIRETORIA COLEGIADA - RDC N° 130, DE 2 DE DEZEMBRO DE 2016" (in Portuguese). Agência Nacional de Vigilância Sanitária (ANVISA) [Brazilian Health Regulatory Agency (ANVISA)]. December 5, 2016.
- ↑ "Schedule III". Controlled Drugs and Substances Act (CDSA). Isomer Design. Retrieved October 10, 2020.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 "2C-I: Legal Status". Erowid. Retrieved March 13, 2014.
- ↑ "Gesetz über den Verkehr mit Betäubungsmitteln: Anlage I" (in German). Bundesamt für Justiz [Federal Office of Justice]. Retrieved December 10, 2019.
- ↑ "Dreizehnte Verordnung zur Änderung betäubungsmittelrechtlicher Vorschriften" (PDF). Bundesgesetzblatt Jahrgang 1999 Teil I Nr. 46 (in German). Bundesanzeiger Verlag [Federal Gazette] (published September 30, 1999). September 24, 1999. p. 1935. eISSN 0344-7634.
- ↑ "Gesetz über den Verkehr mit Betäubungsmitteln: § 29" (in German). Bundesamt für Justiz [Federal Office of Justice]. Retrieved December 10, 2019.
- ↑ "Noteikumi par Latvijā kontrolējamajām narkotiskajām vielām, psihotropajām vielām un prekursoriem" (in Latvian). VSIA Latvijas Vēstnesis. November 10, 2005. Retrieved January 1, 2020.
- ↑ "Föreskrifter om ändring i Läkemedelsverkets föreskrifter (LVFS 1997:12) om förteckningar över narkotika" (PDF) (in Swedish). Läkemedelsverket [Medical Products Agency ] (published March 16, 2004). February 24, 2004. ISSN 1101-5225. LVFS 2004:3. Archived from the original (PDF) on June 13, 2018.
- ↑ "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien" (in German). Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved January 1, 2020.
- ↑ Bakanlar Kurulu Kararı Karar Sayısı : 2013/4827 | https://free-ankara.org/wp-content/uploads/2017/09/BKK_2013_4827_28688.pdf
- ↑ "Schedule 2: Part I: Class A Drugs". "Misuse of Drugs Act 1971". UK Government. Retrieved August 20, 2020.