2C-C
| Summary sheet: 2C-C |
| 2C-C | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chemical Nomenclature | |||||||||||||||||||||||||||||||||
| Common names | 2C-C | ||||||||||||||||||||||||||||||||
| Substitutive name | 4-Chloro-2,5-dimethoxyphenethylamine | ||||||||||||||||||||||||||||||||
| Systematic name | 1-(4-Chloro-2,5-dimethoxyphenyl)-2-aminoethane | ||||||||||||||||||||||||||||||||
| Class Membership | |||||||||||||||||||||||||||||||||
| Psychoactive class | Psychedelic | ||||||||||||||||||||||||||||||||
| Chemical class | Phenethylamine | ||||||||||||||||||||||||||||||||
| Routes of Administration | |||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||
| Interactions | |||||||||||||||||||||||||||||||||
| Cannabis | |||||||||||||||||||||||||||||||||
| Stimulants | |||||||||||||||||||||||||||||||||
| Tramadol | |||||||||||||||||||||||||||||||||
| Lithium | |||||||||||||||||||||||||||||||||
4-Chloro-2,5-dimethoxyphenethylamine (commonly known as 2C-C) is a lesser-known psychedelic substance of the phenethylamine class that produces psychedelic effects when administered.[1] It is a member of the 2C-x family of psychedelic phenethylamines, all of which were derived from the systematic modification of the mescaline molecule.
2C-C was first synthesized by Alice C. Cheng and Neal Castagnoli Jr. in 1983 as an intermediate in a study evaluating the neurotoxicity of 6-hydroxydopamine analogs.[2] Its activity in humans was later investigated and documented by Alexander Shulgin in his book PiHKAL ("Phenethylamines I Have Known and Loved").
Many users report that the effects of 2C-C are gentler, more relaxed, and sedating than other closely related psychedelic phenethylamines such as 2C-B, 2C-I, and 2C-E. It is one of the least potent members of the 2C-x family, with muted visual effects and a relatively unaltered headspace.
Very little data exists about the pharmacological properties, metabolism, and toxicity of 2C-C, and its history of human use is limited. Today, it is used for recreational and therapeutic purposes. It is rarely sold on the streets and distributed online as a grey area research chemical.
Chemistry
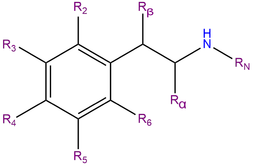
2C-C or 2,5-dimethoxy-4-chlorophenethylamine is a substituted phenethylamine featuring a phenyl ring bound to an amino (NH2) group through an ethyl chain. 2C-C contains methoxy functional groups CH3O- attached to carbons R2 and R5 as well as a chlorine atom attached to carbon R4 of the phenyl ring. 2C-C belongs to the 2C family of phenethylamines which contain methoxy groups on the 2 and 5 positions of the benzene ring.[1]
Pharmacology
2C-C's psychedelic effects are believed to come from its efficacy at the 5-HT2A receptor as a partial agonist. However, the role of these interactions and how they result in the psychedelic experience is the subject of ongoing scientific investigation.
Subjective effects
Disclaimer: The effects listed below cite the Subjective Effect Index (SEI), an open research literature based on anecdotal user reports and the personal analyses of PsychonautWiki contributors. As a result, they should be viewed with a healthy degree of skepticism.
It is also worth noting that these effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects. Likewise, adverse effects become increasingly likely with higher doses and may include addiction, severe injury, or death ☠.
Physical effects 
-
- Stimulation & Sedation - In terms of its effects on the physical energy levels of the user, the 2C-C experience typically begins with mild stimulation, but usually becomes somewhat sedating as the experience progresses and surprisingly lacks the distinctive energetic feeling associated with most phenethylamines. This is described in PiHKAL as "an intense form of relaxation."[1]
- Spontaneous physical sensations - The "body high" of 2C-C can be described as a pleasurable, warm, soft, all-encompassing and mild tingling sensation. This maintains a consistent presence that steadily rises with the onset and hits its limit once the peak has been reached.
- Bodily control enhancement - Whilst at lower doses one may find that their bodily control is enhanced, at higher dosages this becomes suppressed.
- Increased blood pressure
- Increased bodily temperature
- Increased heart rate - This compound seems to not increase heart rate as much as related substances.
- Increased perspiration
- Muscle cramps - Very high dosages of 2C-C are known to cause random muscle tightening, which can become uncomfortable.
- Nausea - Although nausea remains present, in comparison to other phenethylamines such as 2C-E, 2C-I or 2C-B, this remains relatively mild and tends to dissipate quickly. It is considered by some to be one of the most physically understated psychedelics known.
- Dehydration
- Pupil dilation
- Tactile enhancement
- Teeth grinding - This component can be considered to be less intense when compared with that of MDMA.
Visual effects 
-
Enhancements
Distortions
- Drifting (melting, flowing, breathing and morphing) - In comparison to other psychedelics, this effect can be described as highly detailed, slow and smooth in motion, static in appearance and cartoon-like in style.
- Colour shifting
- Tracers
Geometry
The visual geometry of 2C-C is more similar in appearance to that of 2C-B than that of Psilocin, 2C-E, or ayahuasca. They can be comprehensively described as unstructured in their organization, algorithmic in geometric style, intricate in complexity, large, fast and smooth in motion, colorful in scheme, glossy in color, sharp in their edges and angular in their corners.
Hallucinatory states
2C-C can produce a full range of hallucinatory states in a fashion that is more consistent and reproducible than that of many other commonly used psychedelics. However, the dose required to reach this state is significantly higher than many related 2C-x phenethylamines. These effects include:
- Transformations
- Internal hallucination (autonomous entities; settings, sceneries, and landscapes; perspective hallucinations and scenarios and plots) - 2C-C is capable of producing high levels of hallucinogenic imagery, although typically only at very heavy doses. When this effect does occur, it commonly contains hallucinations with scenarios, settings, concepts and autonomous entity contact. They are more common within dark environments and can be described as internal in their manifestation, lucid in believability, interactive in style and almost exclusively of a personal, religious, spiritual, science-fiction, fantasy, surreal, nonsensical or transcendental nature in theme.
- External hallucination (autonomous entities; settings, sceneries, and landscapes; perspective hallucinations and scenarios and plots) - 2C-C is capable of external hallucinations embedded within visual geometry. This particular effect commonly contains hallucinations with scenarios, settings, concepts and autonomous entity contact. They can be described as external in their manifestation, lucid in believability, interactive in style and almost exclusively of a personal, religious, spiritual, science-fiction, fantasy, surreal, nonsensical or transcendental nature in their overall theme.
Cognitive effects 
-
The head space of 2C-C is described by many as one which is both insightful and relatively normal in its thought processes even at moderate to high dosages.
- Empathy, affection, and sociability enhancement - This component is consistently manifested only in the context of social settings in which one is within the company of others. These feelings of sociability, love and empathy are not as pronounced than those found on substances such as MDMA or 2C-B.
- Analysis enhancement - This introspection dominant effect is only manifested consistently in the context of a non-social setting in which the user is alone.
- Conceptual thinking
- Emotion enhancement
- Immersion enhancement
- Increased music appreciation
- Increased libido
- Increased sense of humor
- Increased music appreciation
- Memory suppression - Relative to other psychedelics, 2C-C does not produce this effect except at very high doses.
- Ego death - Relative to other psychedelics, 2C-C does not produce this effect except at very high doses.
- Mindfulness
- Novelty enhancement
- Personal bias suppression
- Thought acceleration
- Thought connectivity
- Time distortion
- Wakefulness - Due to it's relaxing nature, some users report being able to sleep after the peak effects subsided.
Auditory effects 
-
- Auditory enhancement
- Auditory distortion - This effect does not typically occur except at higher doses.
- Auditory hallucination - This effect does not typically occur except at higher doses.
Transpersonal effects 
-
- Unity and interconnectedness - In contrast to some other psychedelics, this effect does not typically occur except at very high doses.
Combinational effects
- Cannabis - Cannabis majorly intensifies and extends both the sensory and cognitive effects of 2C-C. Extreme caution should be exercised with this combination as it can also elevate the anxiety, confusion and psychosis risk of cannabis.
- Dissociatives - When combined with dissociatives, the geometry, euphoria, dissociation and hallucinatory effects are often greatly enhanced. Dissociative-induced holes, spaces, and voids while under the influence of 2C-C have significantly more vivid visuals than dissociatives alone. It also results in more intense internal hallucinations and corresponding confusion which can develop into delusions and psychosis.
- Nitrous - Nitrous oxide is commonly used in combination with psychedelics. The two are known to possess powerful cross-synergistic effects, including the capacity to send the user directly into an "ego death" state. The speed and intensity with which this occurs is very rapid and the euphoria that can result often leads to the urge to compulsively redose.
- MDMA - When combined with MDMA, the physical and cognitive effects of 2C-C become strongly amplified. The visual, physical and cognitive effects of 2C-C are also intensified with strong sensations of euphoric pleasure manifested through distinct body highs and headspaces, and uniquely colorful visuals. The synergy between these substances is unpredictable, and it is best to start with markedly lower dosages than one would take for both substances individually. This combination may increase the neurotoxic effects of MDMA based on its similarity to LSD, which has been found to increase MDMA neurotoxicity.[3]
- Alcohol - Alcohol can increase the disinhibiting and euphoric effects of 2C-C which lends to its use in recreational settings. It can be used in light doses to "take the edge off" a trip as well as dull its psychedelic effects in a fashion somewhat similar to benzodiazepines. However, this is not typically recommended due to alcohol’s ability to cause dehydration and nausea and physical fatigue which can negatively affect a trip if taken in moderate to high dosages. Heavy drinking is strongly discouraged as it can easily lead to black outs and unpredictable behavior.
- Benzodiazepines - Benzodiazepines can, depending on the dosage, slightly to completely reduce the intensity of the cognitive, physical and visual effects of a 2C-C trip. They are very efficient at stopping "bad trips" at the cost of amnesia and reduced trip intensity. Caution is advised when acquiring them for this purpose due to their very high abuse and addiction potential.
- Psychedelics - When used in combination with other psychedelics, each substance's physical, cognitive and visual effects intensify and synergize strongly. The synergy between those substances is unpredictable, and for this reason generally not advised. If choosing to combine psychedelics, it is recommended to start with significantly lower dosages than one would take for either substance individually.
Experience reports
Anecdotal reports which describe the effects of this compound within our experience index include:
Additional experience reports can be found here:
Toxicity and harm potential
The toxicity and long-term health effects of recreational 2C-C use do not appear to have been studied in any scientific context and the exact toxic dose is unknown. This is because 2C-C is a research chemical with a limited history of human use.
Anecdotal reports from those who have tried this compound suggest that there are no negative health effects attributed to simply trying the substance by itself at low to moderate doses and using it very sparingly (although nothing can be completely guaranteed). Independent research should always be conducted to ensure that a combination of two or more substances is safe before consumption.
It is strongly recommended that one use harm reduction practices when using this substance.
Tolerance and addiction potential
Although no formal studies have been conducted, it is not unreasonable to assume that like psychedelics in general, 2C-C is not habit-forming and that the desire to use it can actually decrease with use.
Tolerance to the effects of 2C-C is not built almost immediately after ingestion. Similar to 2C-B there seems to be no apparent tolerance buildup if used repeatedly for a few days. 2C-C does however produce cross-tolerance with other serotonergic psychedelics, meaning that after the use of 2C-C all psychedelics will have a reduced effect.
Dangerous interactions
Warning: Many psychoactive substances that are reasonably safe to use on their own can suddenly become dangerous and even life-threatening when combined with certain other substances. The following list provides some known dangerous interactions (although it is not guaranteed to include all of them).
Always conduct independent research (e.g. Google, DuckDuckGo, PubMed) to ensure that a combination of two or more substances is safe to consume. Some of the listed interactions have been sourced from TripSit.
- Lithium - Lithium is commonly prescribed for the treatment of bipolar disorder. There is a large body of anecdotal evidence that suggests taking it with psychedelics significantly increases the risk of psychosis and seizures. As a result, this combination is strictly discouraged.
- Cannabis - Cannabis may have an unexpectedly strong and unpredictable synergy with the effects of 2C-C. Caution is advised with this combination as it can significantly increase the risk of adverse psychological reactions like anxiety, paranoia, panic attacks, and psychosis. Users are advised to start off with only a fraction of their normal cannabis dose and take long breaks between hits to avoid unintentional overdose.
- Stimulants - Stimulants like amphetamine, cocaine or methylphenidate affect many parts of the brain and alter dopaminergic function. This combination can increase the risk of anxiety, paranoia, panic attacks, and thought loops. This interaction may also result in an elevated risk of mania and psychosis.[citation needed]
- Tramadol - Tramadol is well-documented to lower the seizure threshold[4] and psychedelics may act to trigger seizures in susceptible individuals.[citation needed]
Legal status
- Australia: Australia has a blanket ban over all substituted phenethylamines including the entire 2C-X family.[5]
- Austria: 2C-C is illegal to possess, produce and sell under the NPSG (Neue-Psychoaktive-Substanzen-Gesetz Österreich). In its Schedule II, the further specifying NPSV (Neue-Psychoaktive-Substanzen-Verordnung Österreich) explicitly bans all substituted phenetylamines.[6]
- Brazil: Possession, production and sale is illegal as it is listed on Portaria SVS/MS nº 344.[7]
- Canada: 2C-C would be considered Schedule III as it is a derivative of 2,5-dimethoxyphenethylamine.[8]
- China: As of October 2015, 2C-C is a controlled substance in China.[9]
- Germany: 2C-C is controlled under Anlage I BtMG (Narcotics Act, Schedule I)[10] as of December 13, 2014.[11] It is illegal to manufacture, possess, import, export, buy, sell, procure or dispense it without a license.[12]
- Japan: 2C-C is controlled by the Pharmaceutical Affairs Law in Japan, making it illegal to possess or sell.[13]
- Latvia: 2C-C is a Schedule I controlled substance in Latvia.[14]
- Sweden: 2C-C is a controlled substance.[15]
- Switzerland: 2C-C is a controlled substance specifically named under Verzeichnis E.[16]
- Turkey: 2C-C is classed as a drug and is illegal to possess, produce, supply, or import.[17]
- United Kingdom: 2C-C is a Class A drug in the United Kingdom as a result of the phenethylamine catch-all clause.[18]
- United States: As of July 9, 2012, 2C-C is a Schedule I substance in the United States under the Food and Drug Administration Safety and Innovation Act of 2012, making possession, distribution and manufacture illegal without a DEA license.[19]
See also
External links
Discussion
- The Big & Dandy 2C-C Thread (Bluelight)
- 2C-C, broken down and described (Disregard Everything I Say)
References
- ↑ 1.0 1.1 1.2 Alexander Shulgin; Ann Shulgin (1991). "#22. 2C-C". PiHKAL: A Chemical Love Story. United States: Transform Press. ISBN 0963009605. OCLC 1166889264.
- ↑ Cheng, A. C.; Castagnoli Jr, N. (1984). "Synthesis and physicochemical and neurotoxicity studies of 1-(4-substituted-2, 5-dihydroxyphenyl)-2-aminoethane analogs of 6-hydroxydopamine". Journal of Medicinal Chemistry. 27 (4): 513–520. doi:10.1021/jm00370a014. eISSN 1520-4804. ISSN 0022-2623. OCLC 39480771. PMID 6423824.
- ↑ Armstrong, B. D.; Paik, E.; Chhith, S.; Lelievre, V.; Waschek, J. A.; Howard, S. G. (October 26, 2004). "Potentiation of (DL)‐3, 4‐methylenedioxymethamphetamine (MDMA)‐induced toxicity by the serotonin 2A receptior partial agonist d‐lysergic acid diethylamide (LSD), and the protection of same by the serotonin 2A/2C receptor antagonist MDL 11,939". Neuroscience Research Communications. 35 (2): 83–95. doi:10.1002/nrc.20023. eISSN 1520-6769.
- ↑ Talaie, H.; Panahandeh, R.; Fayaznouri, M. R.; Asadi, Z.; Abdollahi, M. (2009). "Dose-independent occurrence of seizure with tramadol". Journal of Medical Toxicology. 5 (2): 63–67. doi:10.1007/BF03161089. ISSN 1556-9039.
- ↑ "Psychoactive Substances" (PDF). National Drug and Alcohol Research Centre. 2014.[dead link]
- ↑ "Bundesrecht konsolidiert: Gesamte Rechtsvorschrift für Neue-Psychoaktive-Substanzen-Verordnung" (in German). June 26, 2019. Retrieved January 10, 2021.
- ↑ "RESOLUÇÃO DA DIRETORIA COLEGIADA - RDC N° 130, DE 2 DE DEZEMBRO DE 2016" (in Portuguese). Agência Nacional de Vigilância Sanitária (ANVISA) [Brazilian Health Regulatory Agency (ANVISA)]. December 5, 2016.
- ↑ "Schedule III". Controlled Drugs and Substances Act (CDSA). Isomer Design. Retrieved October 10, 2020.
- ↑ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). 国家食品药品监督管理总局 [China Food and Drug Administration (CFDA)]. September 27, 2015. Archived from the original on September 6, 2017.
- ↑ "Gesetz über den Verkehr mit Betäubungsmitteln: Anlage I" (in German). Bundesamt für Justiz [Federal Office of Justice]. Retrieved December 10, 2019.
- ↑ "Achtundzwanzigste Verordnung zur Änderung betäubungsmittelrechtlicher Vorschriften" (PDF). Bundesgesetzblatt Jahrgang 2014 Teil I Nr. 57 (in German). Bundesanzeiger Verlag (published December 12, 2014). December 5, 2014. p. 1999-2002. ISSN 0341-1095. OCLC 231871244.
- ↑ "Gesetz über den Verkehr mit Betäubungsmitteln: § 29" (in German). Bundesamt für Justiz [Federal Office of Justice]. Retrieved December 10, 2019.
- ↑ Uchiyama, N.; Kawamura, M.; Kamakura, H.; Kikura-Hanajiri, R.; Goda, Y. (2008). "Analytical Data of Designated Substances (Shitei-Yakubutsu) Controlled by the Pharmaceutical Affairs Law in Japan, Part I: GC-MS and LC-MS". Yakugaku Zasshi (in Japanese). 128 (6): 971–979. doi:10.1248/yakushi.128.981. eISSN 0031-6903. ISSN 1347-5231. OCLC 909890652. PMID 18520145.
- ↑ "Noteikumi par Latvijā kontrolējamajām narkotiskajām vielām, psihotropajām vielām un prekursoriem" (in Latvian). VSIA Latvijas Vēstnesis. November 10, 2005. Retrieved January 1, 2020.
- ↑ "Svensk författningssamling Förordning om ändring i förordningen (1999:58) om förbud mot vissa hälsofarliga varor" (PDF). Läkemedelsverkets författningssamling (LVFS) (in Swedish). Läkemedelsverket [Swedish Medical Products Agency] (published February 15, 2005). February 3, 2005. ISSN 1101-5225. SFS 2005:26.
- ↑ "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien" (in German). Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved January 1, 2020.
- ↑ Bakanlar Kurulu Kararı Karar Sayısı : 2013/4827 | https://free-ankara.org/wp-content/uploads/2017/09/BKK_2013_4827_28688.pdf
- ↑ "Schedule 2: Part I: Class A Drugs". "Misuse of Drugs Act 1971". UK Government. Retrieved August 20, 2020.
- ↑ "S. 3187 (112th): Food and Drug Administration Safety and Innovation Act". GovTrack. June 27, 2012. Retrieved October 10, 2020.