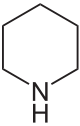
Diphenidine
| Summary sheet: Diphenidine |
| Diphenidine | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chemical Nomenclature | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Common names | Diphenidine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systematic name | (±)-1-(1,2-Diphenylethyl)piperidine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Class Membership | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Psychoactive class | Dissociative | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemical class | Diarylethylamine | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Routes of Administration | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interactions | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stimulants | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Depressants | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Diphenidine (also known as DPD, DND, 1,2-DEP, and DPH) is a lesser-known novel dissociative substance of the diarylethylamine class. It is structurally similar to methoxphenidine (MXP) and ephenidine. It is classified as an NMDA receptor antagonist.[1]
The original synthesis of diphenidine was first reported in 1924.[citation needed] However, it was not selected for further development. Shortly after the 2013 UK arylcyclohexylamine ban, diphenidine and the related compound methoxphenidine became available on the grey market. In 2014, there were two cases of diphenidine being sold in combination with synthetic cannabinoids in Japanese herbal incense blends, one of which was implicated in a fatal overdose.
Subjective effects include stimulation, motor control loss, pain relief, internal hallucinations, conceptual thinking, euphoria, and dissociation. Dissociation is a complex mental state characterized by perceptual distortions and feelings of detachment from the environment and one's self. Anecdotal reports describe high doses of diphenidine producing "bizarre somatosensory phenomena and transient anterograde amnesia."[2] The effects of diarylethylamines share similarities with arylcyclohexylamines like ketamine and phencyclidine (PCP), as well as dextromethorphan (DXM).
Very little data exists about the pharmacological properties, metabolism, and toxicity of diphenidine in humans, and it has an extremely limited history of human usage. Some reports suggest that it may pose different toxicity risks than traditional dissociatives. It is also likely to have moderate to high abuse potential. As a result, it is highly advised to use harm reduction practices if using this substance.
History and culture
This History and culture section is a stub. As a result, it may contain incomplete or wrong information. You can help by expanding it. |
The synthesis of diphenidine was first reported in 1924. It employed a nitrile displacement reaction analogous to the one that would later be used to discover phencyclidine in 1956. Shortly after the 2013 UK ban on arylcyclohexylamines, diphenidine and the related compound methoxphenidine became available on the grey market.
In 2014, there were two cases of diphenidine being sold in combination with synthetic cannabinoids in Japanese herbal incense blends. One herbal incense blend was found to contain diphenidine and 5-fluoro-AB-PINACA at concentrations of 289 mg/g and 55.5 mg/g, respectively.[3] Another product containing AB-CHMINACA, 5F-AMB, and diphenidine was implicated in a fatal overdose.[4]
Names
Diphenidine is sometimes referred to as "DPH" in scientific studies despite this name already being in use and widely accepted as meaning diphenhydramine, an unrelated substance.
Chemistry
Diphenidine is a molecule of the diarylethylamine class. The basic skeleton of diphenidine is a phenethylamine with a substituted Rα in phenyl ring. At the terminals of the piperidine ring, an amino group is attached through phenethylamine chain. Hence, diphenidine belongs to the piperidine dissociative class of compounds. Diphenidine is structurally analogous to MXP, lacking a 2-methoxy substitution on one of its phenyl rings.
Pharmacology
Diphenidine acts as an NMDA receptor antagonist.[5][6] NMDA receptors allow for electrical signals to pass between neurons in the brain and spinal column; for the signals to pass, the receptor must be open. Dissociatives close the NMDA receptors by blocking them. This disconnection of neurons leads to loss of feeling, difficulty moving, and eventually the famous “hole”.
Although vendors of diphenidine have stated the compound acts as a dopamine-reuptake inhibitor and a serotonin reuptake inhibitor with µ-opioid affinity and typical dissociative effects, to date diphenidine has not been screened for affinity at the dopamine transporter. If this is indeed the case, however, it provides an explanation for its euphoric and often stimulating effects.
Diphenidine and related diarylethylamines have been studied in vitro as treatments for neurotoxic injury.[7] Diphenidine may be a stronger NMDA receptor antagonist for neurogenesis, neurological repair and neuroprotection than other more common NMDA receptor antagonistic dissociatives such as ketamine, dextromethorphan, PCP analogs, Iboga and methoxetamine.
Subjective effects
The general head space of diphenidine is often described as euphoric and clear-headed in comparison to that of ketamine and reminiscent of ephenidine at lower dosages. Moderate or higher dosages can sometimes unwillingly turn very confusing and dysphoric without any apparent cause.
Diphenidine is reported to have a much more rapid onset and lower half-life when vaporized or smoked. When consumed this way, it is a suspected to be carcinogenic when excess heat is used. Some user reports have concluded that vaporization requires as low as 20% of what would be a common oral dose for that person.
Disclaimer: The effects listed below cite the Subjective Effect Index (SEI), an open research literature based on anecdotal user reports and the personal analyses of PsychonautWiki contributors. As a result, they should be viewed with a healthy degree of skepticism.
It is also worth noting that these effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects. Likewise, adverse effects become increasingly likely with higher doses and may include addiction, severe injury, or death ☠.
Physical effects 
-
- Stimulation
- Perception of bodily lightness - This creates the sensation that the body is floating and has become entirely weightless. This effect is strangely stimulating and encourages physical activities at low to moderate dosages by making the body feel light and effortless to move.
- Changes in felt bodily form
- Spatial disorientation
- Spontaneous bodily sensations - The diphenidine "body high" is a sharp, pleasurable tingling sensation which is location specific to the hands, feet and head.
- Tactile suppression - This partially to entirely suppresses one's own sense of touch, creating feelings of numbness within the extremities. It is responsible for the anaesthetic properties of this substance.
- Physical autonomy
- Increased salivation
- Increased heart rate
- Motor control loss - A loss of gross and fine motor control alongside of balance and coordination is prevalent within diphenidine and becomes especially strong at higher dosages. This means that one should be sitting down before the onset (unless one is experienced) in case of falling over and injuring oneself.
- Gait alteration
- Euphoria - This results in feelings of physical euphoria which range between mild pleasure to moderate all-encompassing bliss.
- Dizziness - Although uncommon, some people report dizziness under the influence of diphenidine.
- Nausea - It's worth noting that high dose diphenidine trips can sometimes result in nausea and vomiting at the peak of the trip. For most people, this is surprisingly not as unpleasant as they would initially expect due to the accompanying detachment from the physical senses.
- Orgasm suppression & Orgasm enhancement - Orgasm enhancement can sometimes also be present, even at higher doses, although this effect is not reliable.
Visual effects 
-
Suppression
- Visual acuity suppression
- Double vision - This component is prevalent at moderate to heavy dosages and makes reading impossible unless one closes an eye.
- Pattern recognition suppression - This effect generally occurs at higher dosages and makes one unable to recognize and interpret perceivable visual data.
- Frame rate suppression
Distortions
Geometry
The visual geometry produced by diphenidine can be described as very dark and bland when compared to that of ketamine or DXM and often consists of many tiny interlocking and woven lines. It does not extend beyond level 4 and can be comprehensively described through its variations as simplistic in complexity, algorithmic in style, synthetic in feel, unstructured in organization, dimly lit in lighting, multicoloured in scheme, glossy in shading, soft in edges, small in size, slow in speed, smooth in motion, equal in rounded and angular corners, immersive in depth and consistent in intensity.
Hallucinatory states
At high dosages, diphenidine can produce a full range of high level hallucinatory states in a fashion that is less consistent and reproducible than that of many other commonly used psychedelics. These effects include:
- Internal hallucination (autonomous entities; settings, sceneries, and landscapes; perspective hallucinations and scenarios and plots) - In comparison to other dissociatives, this effect can occur at heavy dosages, but is considerably less common than the same effect found within psychedelics and deliriants. It can be comprehensively described through its variations as delirious in believability, fixed in style, equal in new experiences and memory replays in content, autonomous in controllability and solid in style.
Cognitive effects 
-
The specific cognitive effects can be broken down into several separate subcomponents which are listed and described below:
- Anxiety suppression
- Disinhibition
- Depersonalization
- Derealization
- Creativity enhancement
- Dream potentiation
- Memory suppression
- Ego inflation
- Thought deceleration
- Increased music appreciation
- Analysis suppression
- Time distortion
- Cognitive euphoria
- Introspection
- Déjà vu
- Conceptual thinking
- Compulsive redosing
- Amnesia
- Increased libido - This is reported to be experienced at low to mid dosage ranges.
Disconnective effects 
-
- Tactile disconnection
- Consciousness disconnection
- Visual disconnection - This eventually results in the diphenidine's equivalent of the "K-hole" or more specifically, holes, spaces and voids alongside of structures.
Experience reports
Anecdotal reports which describe the effects of this compound within our experience index include:
- Experience:110mg Diphenidine (vaporized) + 354mg DXM - instant ego death
- Experience:110mg Diphenidine (vaporized) + 354mg DXM- instant ego death
- Experience:300μg 1P-LSD + 40mg diphenidine - My first psychotic break
- Experience:50mg - Diphenidine ride
- Experience:65mg + 30mg redose - My first experience with diphenedine
Additional experience reports can be found here:
Toxicity and harm potential
 |
This toxicity and harm potential section is a stub. As a result, it may contain incomplete or even dangerously wrong information! You can help by expanding upon or correcting it. |
The toxicity and long-term health effects of recreational diphenidine use do not appear to have been studied in any scientific context and the exact toxic dosage is unknown. This is because diphenidine has very little history of human usage.
Some anecdotal reports suggest diphenidine may increase the risk of mania and psychosis. This is common with many dissociatives, particularly with those that provide stimulation such as PCP.
It has been reported that regular use can lead to increased blood pressure and rapid heart rate.
It is strongly recommended that one use harm reduction practices when using this substance.
Dependence and abuse potential
As with other NMDA receptor antagonists, the chronic use of diphenidine can be considered moderately addictive with a high potential for abuse. It is likely capable of causing psychological dependence among certain users. When addiction has developed, cravings and withdrawal effects may occur if one suddenly stops their usage.
Tolerance to many of the effects of diphenidine develops with prolonged and repeated use. This results in users having to administer increasingly large doses to achieve the same effects. After that, it takes about 3 - 7 days for the tolerance to be reduced to half and 1 - 2 weeks to be back at baseline (in the absence of further consumption). Diphenidine presents cross-tolerance with all dissociatives, meaning that after the consumption of diphenidine all dissociatives will have a reduced effect.
Dangerous interactions
Warning: Many psychoactive substances that are reasonably safe to use on their own can suddenly become dangerous and even life-threatening when combined with certain other substances. The following list provides some known dangerous interactions (although it is not guaranteed to include all of them).
Always conduct independent research (e.g. Google, DuckDuckGo, PubMed) to ensure that a combination of two or more substances is safe to consume. Some of the listed interactions have been sourced from TripSit.
- Stimulants - Both stimulants and dissociatives carry the risk of adverse psychological reactions like anxiety, mania, delusions and psychosis and these risks are exacerbated when the two substances are combined.
- Depressants - Because both depress the respiratory system, this combination can result in an increased risk of suddenly falling unconscious, vomiting and choking to death from the resulting suffocation. If nausea or vomiting occurs, users should attempt to fall asleep in the recovery position or have a friend move them into it.
Legal status
Diphenidine is currently a legal grey area drug worldwide and is easily accessible through the use of online research chemical vendors. However, this does not guarantee anyone to be immune from legal prosecution should they be found in possession of this substance as the legality is likely to vary from country to country.
- Canada: As of March 2016, MT-45 and its analogues, one of which being Diphenidine, are Schedule I controlled substances.[8] Possession without legal authority can result in maximum 7 years imprisonment. Only those with a law enforcement agency, person with an exemption permit or institutions with Minister's authorization may possess the drug.
- Switzerland: Diphenidine is a controlled substance specifically named under Verzeichnis E.[9]
- Turkey: Diphenidine is a classed as drug and is illegal to possess, produce, supply, or import.[10]
- United Kingdom: It is illegal to produce, supply, or import this drug under the Psychoactive Substance Act, which came into effect on May 26th, 2016.[11]
- United States: Diphenidine is a schedule II substance and is illegal without a prescription.[12]
See also
External links
Community
Literature
- Wallach, J., Kang, H., Colestock, T., Morris, H., Bortolotto, Z. A., Collingridge, G. L., ... & Adejare, A. (2016). Pharmacological investigations of the dissociative ‘legal highs’ diphenidine, methoxphenidine and analogues. PLoS One, 11(6), e0157021. https://doi.org/10.1371/journal.pone.0157021
- Morris, H., & Wallach, J. (2014). From PCP to MXE: A comprehensive review of the non-medical use of dissociative drugs. Drug Testing and Analysis, 6(7–8), 614–632. https://doi.org/10.1002/dta.1620
References
- ↑ Wallach, J., Kang, H., Colestock, T., Morris, H., Bortolotto, Z. A., Collingridge, G. L., Lodge, D., Halberstadt, A. L., Brandt, S. D., Adejare, A. (17 June 2016). Lee, J., ed. "Pharmacological Investigations of the Dissociative 'Legal Highs' Diphenidine, Methoxphenidine and Analogues". PLOS ONE. 11 (6): e0157021. doi:10.1371/journal.pone.0157021. ISSN 1932-6203.
- ↑ Morris, H., Wallach, J. (July 2014). "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs: PCP to MXE". Drug Testing and Analysis. 6 (7–8): 614–632. doi:10.1002/dta.1620. ISSN 1942-7603.
- ↑ Wurita, A., Hasegawa, K., Minakata, K., Watanabe, K., Suzuki, O. (1 August 2014). "A large amount of new designer drug diphenidine coexisting with a synthetic cannabinoid 5-fluoro-AB-PINACA found in a dubious herbal product". Forensic Toxicology. 32 (2): 331–337. doi:10.1007/s11419-014-0240-y. ISSN 1860-8973.
- ↑ Hasegawa, K., Wurita, A., Minakata, K., Gonmori, K., Nozawa, H., Yamagishi, I., Watanabe, K., Suzuki, O. (1 January 2015). "Postmortem distribution of AB-CHMINACA, 5-fluoro-AMB, and diphenidine in body fluids and solid tissues in a fatal poisoning case: usefulness of adipose tissue for detection of the drugs in unchanged forms". Forensic Toxicology. 33 (1): 45–53. doi:10.1007/s11419-014-0245-6. ISSN 1860-8973.
- ↑ Berger, M. L., Schweifer, A., Rebernik, P., Hammerschmidt, F. (1 May 2009). "NMDA receptor affinities of 1,2-diphenylethylamine and 1-(1,2-diphenylethyl)piperidine enantiomers and of related compounds". Bioorganic & Medicinal Chemistry. 17 (9): 3456–3462. doi:10.1016/j.bmc.2009.03.025. ISSN 0968-0896.
- ↑ Wallach, J., Kavanagh, P. V., McLaughlin, G., Morris, N., Power, J. D., Elliott, S. P., Mercier, M. S., Lodge, D., Morris, H., Dempster, N. M., Brandt, S. D. (May 2015). "Preparation and characterization of the 'research chemical' diphenidine, its pyrrolidine analogue, and their 2,2-diphenylethyl isomers: Characterization of diphenylethylamines". Drug Testing and Analysis. 7 (5): 358–367. doi:10.1002/dta.1689. ISSN 1942-7603.
- ↑ Gray, N. M., Cheng, B. K., 1,2-diarylethylamines for treatment of neurotoxic injury
- ↑ Government of Canada, P. W. and G. S. C. (2016), Canada Gazette – Regulations Amending the Food and Drug Regulations (Parts G and J — Lefetamine, AH-7921, MT-45 and W-18)
- ↑ "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien" (in German). Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved January 1, 2020.
- ↑ https://resmigazete.gov.tr/eskiler/2017/01/20170112-8.pdf
- ↑ Psychoactive Substances Act 2016
- ↑ https://www.unodc.org/LSS/Substance/Details/7ee627dd-c3ed-42d2-b84f-81edef0d6466