Lorazepam

Fatal overdose may occur when benzodiazepines are combined with other depressants such as opiates, barbiturates, gabapentinoids, thienodiazepines, alcohol or other GABAergic substances.[1]
It is strongly discouraged to combine these substances, particularly in common to heavy doses.
| Summary sheet: Lorazepam |
| Lorazepam | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chemical Nomenclature | |||||||||||||||||||||||||||||
| Common names | Lorazepam, Ativan, Orfidal, Lorsilan | ||||||||||||||||||||||||||||
| Substitutive name | Lorazepam | ||||||||||||||||||||||||||||
| Systematic name | (RS)-7-Chloro-5-(2-chlorophenyl)-3-hydroxy-1,3-dihydro-2H-1,4-benzodiazepin-2-one | ||||||||||||||||||||||||||||
| Class Membership | |||||||||||||||||||||||||||||
| Psychoactive class | Depressant | ||||||||||||||||||||||||||||
| Chemical class | Benzodiazepine | ||||||||||||||||||||||||||||
| Routes of Administration | |||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
| Interactions | |||||||||||||||||||||||||||||
Lorazepam (trade name Ativan or Tavor) is a depressant substance of the benzodiazepine class. Lorazepam is used for the short-term treatment of anxiety, insomnia, acute seizures, and the sedation of hospitalized patients.[2][3][4][5]
Users should note that with benzodiazepines, sudden discontinuation can be dangerous or even life-threatening for long-term or heavy users. As a result, individuals who are physically dependent on this substance are advised to taper their dose by gradually lowering the amount taken each day over a prolonged period of time instead of stopping use abruptly.[6]
Chemistry
Lorazepam is a drug of the benzodiazepine class. Benzodiazepine drugs contain a benzene ring fused to a diazepine ring, which is a seven membered ring with the two nitrogen constituents located at R1 and R4. Further, the benzodiazepine ring is bonded at R5 to a 2-chlorinated phenyl ring. R7 of the benzyl ring is also substituted with a chlorine group. Additionally, Lorazepam contains a hydroxy (OH-) group substituted at R3. It also contains an oxygen group double bonded to R2 of its diazepine ring to form a ketone. This oxygen substitution at R2 is shared with other benzodiazepine drugs with the suffix -azepam.
Pharmacology
Benzodiazepines produce a variety of effects by binding to the benzodiazepine receptor site and magnifying the efficiency and effects of the neurotransmitter gamma aminobutyric acid (GABA) by acting on its receptors.[7] As this site is the most prolific inhibitory receptor set within the brain, its modulation results in the sedating (or calming effects) of lorazepam on the nervous system.
The anticonvulsant properties of benzodiazepines may be, in part or entirely, due to binding to voltage-dependent sodium channels rather than benzodiazepine receptors.[8]
Lorazepam is thought to have a relatively high affinity for GABA receptors compared to other benzodiazepines, which may also explain its marked amnesic effects.[9]
Lorazepam undergoes glucuronidation via Hepatic Pathways which produces lorazepam glucuronide as the main metabolite. The glucuronide attached increases the polarity of the molecule allowing a faster excretion through urine.[citation needed]
Subjective effects
Disclaimer: The effects listed below cite the Subjective Effect Index (SEI), an open research literature based on anecdotal user reports and the personal analyses of PsychonautWiki contributors. As a result, they should be viewed with a healthy degree of skepticism.
It is also worth noting that these effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects. Likewise, adverse effects become increasingly likely with higher doses and may include addiction, severe injury, or death ☠.
Physical effects 
-
- Sedation
- Motor control loss
- Muscle relaxation
- Appetite enhancement - This effect is not particularly prominent, but is reported to occur in some people. It can have a synergistic effect when combined with cannabis
- Dizziness
- Nausea suppression[10]
- Respiratory depression
- Seizure suppression
- Frequent urination[11]
Paradoxical effects 
- Paradoxical reactions to benzodiazepines such as increased seizures (in epileptics), aggression, increased anxiety, violent behavior, loss of impulse control, irritability and suicidal behavior sometimes occur (although they are rare in the general population, with an incidence rate below 1%).[12][13] These paradoxical effects occur with greater frequency in recreational abusers, individuals with mental disorders, children, and patients on high-dosage regimes.[14][15]
Cognitive effects 
-
The cognitive effects of lorazepam can be broken down into several components which progressively intensify proportional to dosage. The general head space of lorazepam is described by many as one of intense sedation and decreased inhibition. In recreational doses it can be very discombobulating. It contains a large number of typical depressants cognitive effects. Often compared to alprazolam, lorazepam is noted for having a more sedation and sleepiness but less anxiolytic effects.
The most prominent of these cognitive effects generally include:
- Analysis suppression
- Anxiety suppression - Not as prominent as with alprazolam, clonazepam or diazepam.
- Appetite enhancement
- Compulsive redosing
- Delusions of sobriety - This is the false belief that one is perfectly sober despite obvious evidence to the contrary such as severe cognitive impairment and an inability to fully communicate with others. It most commonly occurs at heavy dosages.
- Disinhibition
- Dream potentiation
- Emotion suppression - Although this compound primarily suppresses anxiety, it also dulls other emotions in a manner which is distinct but less intensive than that of antipsychotics. This particular component may even be more prominent on lorazepam than other common benzodiazepines.
- Memory suppression
- Thought deceleration - This can often be visibly clear to onlookers just during verbal exchanges even on light-medium doses without tolerance.
- Black out
Experience reports
There are currently no anecdotal reports which describe the effects of this compound within our experience index. Additional experience reports can be found here:
Toxicity and harm potential
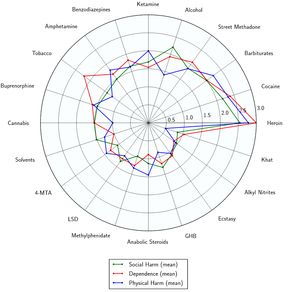
Lorazepam likely has a low toxicity relative to dose.[17] However, it is potentially lethal when mixed with depressants like alcohol or opioids.
It is strongly recommended that one use harm reduction practices when using this substance.
Tolerance and addiction potential
Lorazepam is extremely physically and psychologically addictive.
Tolerance will develop to the sedative-hypnotic effects within a couple of days of continuous use. After cessation, the tolerance returns to baseline in 7 - 14 days. However, in certain cases this may take significantly longer in a manner which is proportional to the duration and intensity of one's long-term usage.
Withdrawal symptoms or rebound symptoms may occur after ceasing usage abruptly following a few weeks or longer of steady dosing, and may necessitate a gradual dose reduction. For more information on tapering from benzodiazepines in a controlled manner, please see this guide.
Benzodiazepine discontinuation is notoriously difficult; it is potentially life-threatening for individuals using regularly to discontinue use without tapering their dose over a period of weeks. There is an increased risk of hypertension, seizures, and death.[18] Drugs which lower the seizure threshold such as tramadol should be avoided during withdrawal.
Lorazepam presents cross-tolerance with all benzodiazepines, meaning that after its consumption all benzodiazepines will have a reduced effect.
Overdose
Benzodiazepine overdose may occur when a benzodiazepine is taken in extremely heavy quantities or concurrently with other depressants. This is particularly dangerous with other GABAergic depressants such as barbiturates and alcohol since they work in a similar fashion, but bind to distinct allosteric sites on the GABAA receptor, thus their effects potentiate one another. Benzodiazepines increase the frequency in which the chlorine ion pore opens on the GABAA receptor while barbiturates increase the duration in which they are open, meaning when both are consumed, the ion pore will open more frequently and stay open longer[19]. Benzodiazepine overdose is a medical emergency that may lead to a coma, permanent brain injury or death if not treated promptly and properly.
Symptoms of a benzodiazepine overdose may include severe thought deceleration, slurred speech, confusion, delusions, respiratory depression, coma or death. Benzodiazepine overdoses may be treated effectively in a hospital environment, with generally favorable outcomes. Benzodiazepine overdoses are sometimes treated with flumazenil, a GABAA antagonist[20], however care is primarily supportive in nature.
Dangerous interactions
Although many drugs are safe on their own, they can become dangerous and even life-threatening when combined with other substances. The list below contains some common potentially dangerous combinations, but may not include all of them. Certain combinations may be safe in low doses of each but still increase the potential risk of death. Independent research should always be done to ensure that a combination of two or more substances is safe before consumption.
- Depressants (1,4-Butanediol, 2-methyl-2-butanol, alcohol, barbiturates, GHB/GBL, methaqualone, opioids) - This combination can result in dangerous or even fatal levels of respiratory depression. These substances potentiate the muscle relaxation, sedation and amnesia caused by one another and can lead to unexpected loss of consciousness at high doses. There is also an increased risk of vomiting during unconsciousness and death from the resulting suffocation. If this occurs, users should attempt to fall asleep in the recovery position or have a friend move them into it.
- Dissociatives - This combination can result in an increased risk of vomiting during unconsciousness and death from the resulting suffocation. If this occurs, users should attempt to fall asleep in the recovery position or have a friend move them into it.
- Stimulants - It is dangerous to combine benzodiazepines with stimulants due to the risk of excessive intoxication. Stimulants decrease the sedative effect of benzodiazepines, which is the main factor most people consider when determining their level of intoxication. Once the stimulant wears off, the effects of benzodiazepines will be significantly increased, leading to intensified disinhibition as well as other effects. If combined, one should strictly limit themselves to only dosing a certain amount of benzodiazepines per hour. This combination can also potentially result in severe dehydration if hydration is not monitored.
Legal status
Internationally, lorazepam is a Schedule IV drug under the United Nations Convention on Psychotropic Substances.[21]
- Austria: Lorazepam is legal for medical use under the AMG (Arzneimittelgesetz Österreich) and illegal when sold or possessed without a prescription under the SMG (Suchtmittelgesetz Österreich).[citation needed]
- Canada: Lorazepam is listed in Schedule IV of the Controlled Drugs and Substances Act in Canada.[citation needed]
- China: Lorazepam is a controlled Class II psychotropic substances.[22] Prescriptions for psychotropic substances in Class II are generally limited to a 7-day supply.[23]
- Germany: Lorazepam is controlled under Anlage III BtMG (Narcotics Act, Schedule III)[24] as of August 1, 1986.[25] It can only be prescribed on a narcotic prescription form, except preparations which contain up to 2.5 mg lorazepam in each dosage form.[24]
- New Zealand: Lorazepam is a Class C Controlled drug under the Misuse of Drugs Act 1975.[26]
- Russia: Lorazepam is a Schedule III controlled substance since 2013.[27]
- Switzerland: Lorazepam is a controlled substance specifically named under Verzeichnis B. Medicinal use is permitted.[28]
- Turkey: Lorazepam is a 'green prescription' only substance[29] and illegal when sold or possessed without a prescription.[citation needed]
- United Kingdom: Lorazepam is classified as a controlled substance and is listed under Schedule IV, Part I (CD Benz POM) of the Misuse of Drugs Regulations 2001, allowing possession with a valid prescription. The Misuse of Drugs Act 1971 makes it illegal to possess it without a prescription and, for such purposes, it is classified as a Class C drug.[30]
- United States: Lorazepam is a Schedule IV drug under the Controlled Substances Act.[31]
- Poland: Lorazepam is controlled under act of psychotropic substances list IV-P Group of "low potential for abuse" (with other benzodiazepines in this group). It's legal for medical, scientific and manufacturing purposes. [32]
See also
- Responsible use
- Psychoactive substance index
- Etizolam
- Benzodiazepines
- Depressants
- Volumetric liquid dosing
External links
- Lorazepam (Wikipedia)
- Lorazepam (Erowid Vault)
- Lorazepam (Isomer Design)
- Lorazepam (DrugBank)
- Lorazepam (Drugs.com)
- Lorazepam (Drugs-Forum)
References
- ↑ Risks of Combining Depressants - TripSit
- ↑ benzo.org.uk : Benzodiazepines and their effects, Professor Ian Hindmarch, January, 1997
- ↑ Cox, C. E., Reed, S. D., Govert, J. A., Rodgers, J. E., Campbell-Bright, S., Kress, J. P., Carson, S. S. (March 2008). "An Economic Evaluation of Propofol and Lorazepam for Critically Ill Patients Undergoing Mechanical Ventilation". Critical care medicine. 36 (3): 706–714. doi:10.1097/CCM.0B013E3181544248. ISSN 0090-3493.
- ↑ Walker, M. (24 September 2005). "Status epilepticus: an evidence based guide". BMJ : British Medical Journal. 331 (7518): 673–677. ISSN 0959-8138.
- ↑ Battaglia, J. (1 June 2005). "Pharmacological Management of Acute Agitation". Drugs. 65 (9): 1207–1222. doi:10.2165/00003495-200565090-00003. ISSN 1179-1950.
- ↑ Kahan, M., Wilson, L., Mailis-Gagnon, A., Srivastava, A. (November 2011). "Canadian guideline for safe and effective use of opioids for chronic noncancer pain. Appendix B-6: Benzodiazepine Tapering". Canadian Family Physician. 57 (11): 1269–1276. ISSN 0008-350X.
- ↑ Haefely, W. (29 June 1984). "Benzodiazepine interactions with GABA receptors". Neuroscience Letters. 47 (3): 201–206. doi:10.1016/0304-3940(84)90514-7. ISSN 0304-3940.
- ↑ McLean, M. J., Macdonald, R. L. (February 1988). "Benzodiazepines, but not beta carbolines, limit high frequency repetitive firing of action potentials of spinal cord neurons in cell culture". The Journal of Pharmacology and Experimental Therapeutics. 244 (2): 789–795. ISSN 0022-3565.
- ↑ Matthew, E., Andreason, P., Pettigrew, K., Carson, R. E., Herscovitch, P., Cohen, R., King, C., Johanson, C. E., Greenblatt, D. J., Paul, S. M. (28 March 1995). "Benzodiazepine receptors mediate regional blood flow changes in the living human brain". Proceedings of the National Academy of Sciences of the United States of America. 92 (7): 2775–2779. ISSN 0027-8424.
- ↑ http://www.drugs.com/comments/lorazepam/for-nausea-vomiting.html
- ↑ Lorazepam: MedlinePlus Drug Information
- ↑ Saïas, T., Gallarda, T. (September 2008). "[Paradoxical aggressive reactions to benzodiazepine use: a review]". L’Encephale. 34 (4): 330–336. doi:10.1016/j.encep.2007.05.005. ISSN 0013-7006.
- ↑ Paton, C. (December 2002). "Benzodiazepines and disinhibition: a review". Psychiatric Bulletin. 26 (12): 460–462. doi:10.1192/pb.26.12.460. ISSN 0955-6036.
- ↑ Bond, A. J. (1 January 1998). "Drug- Induced Behavioural Disinhibition". CNS Drugs. 9 (1): 41–57. doi:10.2165/00023210-199809010-00005. ISSN 1179-1934.
- ↑ Drummer, O. H. (February 2002). "Benzodiazepines - Effects on Human Performance and Behavior". Forensic Science Review. 14 (1–2): 1–14. ISSN 1042-7201.
- ↑ Nutt, D., King, L. A., Saulsbury, W., Blakemore, C. (24 March 2007). "Development of a rational scale to assess the harm of drugs of potential misuse". The Lancet. 369 (9566): 1047–1053. doi:10.1016/S0140-6736(07)60464-4. ISSN 0140-6736.
- ↑ Mandrioli, R., Mercolini, L., Raggi, M. A. (October 2008). "Benzodiazepine metabolism: an analytical perspective". Current Drug Metabolism. 9 (8): 827–844. doi:10.2174/138920008786049258. ISSN 1389-2002.
- ↑ Lann, M. A., Molina, D. K. (June 2009). "A fatal case of benzodiazepine withdrawal". The American Journal of Forensic Medicine and Pathology. 30 (2): 177–179. doi:10.1097/PAF.0b013e3181875aa0. ISSN 1533-404X.
- ↑ Twyman, R. E., Rogers, C. J., Macdonald, R. L. (March 1989). "Differential regulation of gamma-aminobutyric acid receptor channels by diazepam and phenobarbital". Annals of Neurology. 25 (3): 213–220. doi:10.1002/ana.410250302. ISSN 0364-5134.
- ↑ Hoffman, E. J., Warren, E. W. (September 1993). "Flumazenil: a benzodiazepine antagonist". Clinical Pharmacy. 12 (9): 641–656; quiz 699–701. ISSN 0278-2677.
- ↑ List of psychotropic substances under international control | http://www.indiapost.gov.in/Pdf/Customs/List_of_Psychotropic_Substances.pdf
- ↑ 《麻醉药品和精神药品品种目录(2023版)》-国有资产管理处 (tjnu.edu.cn)
- ↑ 卫生部关于印发《麻醉药品、精神药品处方管理规定》的通知 麻醉药品、精神药品处方管理规定__2006年第28号国务院公报_中国政府网 (www.gov.cn)
- ↑ 24.0 24.1 "Anlage III BtMG" (in German). Bundesministerium der Justiz und für Verbraucherschutz. Retrieved December 26, 2019.
- ↑ "Zweite Verordnung zur Änderung betäubungsmittelrechtlicher Vorschriften" (PDF). Bundesgesetzblatt Jahrgang 1986 Teil I Nr. 36 (in German). Bundesanzeiger Verlag. July 29, 1986. Retrieved December 26, 2019.
- ↑ http://legislation.govt.nz/act/public/1975/0116/91.0/DLM436723.html
- ↑ Постановление Правительства РФ от 04.02.2013 N 78 “О внесении изменений в некоторые акты Правительства Российской Федерации” - КонсультантПлюс
- ↑ "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien" (in German). Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved January 1, 2020.
- ↑ YEŞİL REÇETEYE TABİ İLAÇLAR | https://www.titck.gov.tr/storage/Archive/2019/contentFile/01.04.2019%20SKRS%20Ye%C5%9Fil%20Re%C3%A7eteli%20%C4%B0la%C3%A7lar%20Aktif%20SON%20-%20G%C3%9CNCEL_58b1ff4a-2e1c-4867-bad7-eec855d6162a.pdf
- ↑ Drugs licensing
- ↑ Drug Scheduling, DEA | https://www.dea.gov/drug-information/drug-scheduling
- ↑ Polish wiki about controlled substances (no english translation)