Diazepam

Fatal overdose may occur when benzodiazepines are combined with other depressants such as opiates, barbiturates, gabapentinoids, thienodiazepines, alcohol or other GABAergic substances.[1]
It is strongly discouraged to combine these substances, particularly in common to heavy doses.
| Summary sheet: Diazepam |
| Diazepam | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chemical Nomenclature | |||||||||||||||||||||||||||||
| Common names | Valium, Diastat, Mother's Little Helper, Apaurin | ||||||||||||||||||||||||||||
| Substitutive name | Diazepam | ||||||||||||||||||||||||||||
| Systematic name | 7-Chloro-1-methyl-5-phenyl-3H-1,4-benzodiazepin-2-one | ||||||||||||||||||||||||||||
| Class Membership | |||||||||||||||||||||||||||||
| Psychoactive class | Depressant | ||||||||||||||||||||||||||||
| Chemical class | Benzodiazepine | ||||||||||||||||||||||||||||
| Routes of Administration | |||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||
| Interactions | |||||||||||||||||||||||||||||
| Depressants | |||||||||||||||||||||||||||||
| Dissociatives | |||||||||||||||||||||||||||||
| Stimulants | |||||||||||||||||||||||||||||
Diazepam (also known as Valium) is a depressant substance of the benzodiazepine class. Its mechanism of action is to increase the effects of the inhibitory neurotransmitter GABA.
Diazepam was patented in 1955 by the pharmaceutical company Hoffman-La Roche.[2] It has been one of the most frequently prescribed medications in the world since its launch in 1963.[3] It is commonly used to treat a wide range of conditions including anxiety, panic attacks, insomnia, seizures, muscle spasms, and restless legs syndrome. Diazepam is a core medicine in the World Health Organization's Essential Drugs List, the minimum medical needs for a basic health-care system.[4]
Subjective effects include anxiety suppression, sedation, and muscle relaxation.[5] Diazepam has a rapid onset of action compared to other benzodiazepines. However, it is generally considered to have lesser recreational effects compared to benzodiazepines like clonazepam (Klonopin) and alprazolam (Xanax).
Diazepam is considered to have low toxicity.[citation needed] However, it has moderate abuse potential and produces physical and psychological dependence with extended use. Additionally, it can contribute to death by respiratory depression if combined with alcohol, opiates, and other depressants. It is highly advised to use harm reduction practices if using this substance.
It should be noted that the sudden discontinuation of benzodiazepines can be potentially dangerous or life-threatening for individuals using them regularly for extended periods of time, sometimes resulting in seizures or death.[6] It is highly recommended to taper one's dose by gradually lowering the amount taken each day for a prolonged period of time instead of stopping abruptly.[7]
While diazepam itself does not naturally occur in plants or animals, recent research has indicated that low concentrations of its major metabolite, desmethyldiazepam (DD), can be found in various biological systems, including those of animals and certain plants. This raises intriguing questions about the potential for these compounds to enter the food chain and their possible natural origins. However, the clinical significance and biological roles of these findings are still being explored.[8][9]
History and culture
Following chlordiazepoxide (Librium), which was approved for use in 1960, diazepam was the second benzodiazepine invented by Leo Sternbach of pharmaceutical company Hoffman-La Roche. Released in 1963 as an improved version of Librium, diazepam became incredibly popular and quickly surpassed it in sales, helping Roche to become a pharmaceutical industry giant. After this initial success, other pharmaceutical companies began to introduce other benzodiazepine derivatives.[10]
The benzodiazepines gained popularity among medical professionals as an improvement over barbiturates, which have a comparatively narrow therapeutic index, and are far more sedative at therapeutic doses. The benzodiazepines are also far less dangerous; death rarely results from diazepam overdose, except in cases where it is consumed with large amounts of other depressants (such as alcohol or opioids).[11] Benzodiazepine drugs such as diazepam initially had widespread public support, but with time the view changed to one of growing criticism and calls for restrictions on their prescription.[12]
Marketed by Roche using an advertising campaign conceived by the William Douglas McAdams Agency under the leadership of Arthur Sackler,[13] Diazepam was the top-selling pharmaceutical in the United States from 1969 to 1982, with peak annual sales in 1978 of 2.3 billion Valium tablets.[10] While psychiatrists continue to prescribe diazepam for the short-term relief of anxiety, neurology has taken the lead in prescribing diazepam for the palliative treatment of certain types of epilepsy and spastic activity.[citation needed]
Chemistry
Diazepam is a drug of the benzodiazepine class. Benzodiazepine drugs contain a benzene ring fused to a diazepine ring, which is a seven-membered ring with the two nitrogen constituents located at R1 and R4. At R1, diazepam is substituted with methyl group. Further, the benzodiazepine ring is bonded at R5 to an aromatic phenyl ring. The benzyl ring of the bicyclic core is substituted at R7 with a chlorine group. Diazepam also contains an oxygen group double-bonded to R2 of its diazepine ring to form a ketone. This oxygen substitution at R2 is shared with other benzodiazepine drugs with the suffix -azepam.
Diazepam is a 1,4-benzodiazepine. Diazepam occurs as solid white or yellow crystals with a melting point of 131.5 to 134.5 °C. It is odorless, and has a slightly bitter taste. The pH of diazepam is neutral (i.e., pH = 7). Due to additives such as benzoic acid/benzoate in the injectable form. Diazepam can absorb into plastics, so liquid preparations should not be kept in plastic bottles or syringes, etc. As such, it can leach into the plastic bags and tubing used for intravenous infusions. Absorption appears to depend on several factors, such as temperature, concentration, flow rates, and tube length. Diazepam should not be administered if a precipitate has formed and does not dissolve.[14]
Pharmacology
Diazepam is a long-acting "classical" benzodiazepine. Benzodiazepines act via micromolar benzodiazepine binding sites as calcium channel blockers and significantly inhibit depolarization-sensitive calcium uptake in rat nerve cell preparations.[15] Diazepam inhibits acetylcholine release in mouse hippocampal synaptosomes. This has been found by measuring sodium-dependent high-affinity choline uptake in mouse brain cells in vitro, after pretreatment of the mice with diazepam in vivo. This may play a role in explaining diazepam's anticonvulsant properties.[16]
Benzodiazepines are positive allosteric modulators of the GABA type A receptors (GABAA). The GABAA receptors are ligand-gated chloride-selective ion channels that are activated by GABA, the major inhibitory neurotransmitter in the brain. Binding of benzodiazepines to this receptor complex promotes the binding of GABA, which in turn increases the total conduction of chloride ions across the neuronal cell membrane. This increased chloride ion influx hyperpolarizes the neuron's membrane potential. As a result, the difference between resting potential and threshold potential is increased, and firing is less likely. As a result, the arousal of the cortical and limbic systems in the central nervous system is reduced.[17]
The GABAA receptor is a heteromer composed of five subunits, the most common ones being two αs, two βs, and one γ (α2β2γ). For each subunit, many subtypes exist (α1–6, β1–3, and γ1–3). GABAA receptors containing the α1 subunit mediate the sedative, the anterograde amnesic, and partly the anticonvulsive effects of diazepam. GABAA receptors containing α2 mediate the anxiolytic actions and, to a large degree, the myorelaxant effects. GABAA receptors containing α3 and α5 also contribute to benzodiazepines myorelaxant actions, whereas GABAA receptors comprising the α5 subunit were shown to modulate the temporal and spatial memory effects of benzodiazepines.[18]
Diazepam is not the only drug to target these GABAA receptors. Drugs such as flumazenil also bind to GABAA to induce their effects.[19] Diazepam appears to act on areas of the limbic system, thalamus, and hypothalamus, inducing anxiolytic effects. Benzodiazepine drugs, including diazepam increase the inhibitory processes in the cerebral cortex.[20]
Benzodiazepines produce a variety of effects by binding to the benzodiazepine receptor site and magnifying the efficiency and effects of the neurotransmitter gamma aminobutyric acid (GABA) by acting on its receptors.[21] As this site is the most prolific inhibitory receptor set within the brain, its modulation results in the sedating (or calming effects) of diazepam on the nervous system. The anticonvulsant properties of benzodiazepines may be, in part or entirely, due to binding to voltage-dependent sodium channels rather than benzodiazepine receptors.[22]
Subjective effects
Disclaimer: The effects listed below cite the Subjective Effect Index (SEI), an open research literature based on anecdotal user reports and the personal analyses of PsychonautWiki contributors. As a result, they should be viewed with a healthy degree of skepticism.
It is also worth noting that these effects will not necessarily occur in a predictable or reliable manner, although higher doses are more liable to induce the full spectrum of effects. Likewise, adverse effects become increasingly likely with higher doses and may include addiction, severe injury, or death ☠.
Physical effects 
-
- Sedation - In terms of energy level alterations, this drug is sedating and often results in an overwhelmingly lethargic state. At higher levels, this causes users to suddenly feel as if they are extremely sleep deprived and have not slept for days, forcing them to sit down and generally feel as if they are constantly on the verge of passing out instead of engaging in physical activities. This sense of sleep deprivation increases proportional to dosage and eventually becomes powerful enough to force a person into complete unconsciousness.
- Muscle relaxation - In comparison to alprazolam (Xanax), diazepam has greater amounts of muscle relaxation.
- Motor control loss
- Respiratory depression
- Dizziness
- Seizure suppression
- Physical euphoria - Diazepam is similar to other benzodiazepines, where despite suppressing emotion, people report moderate-strong feelings of relaxation, pleasure and comfort in the body. This seems to present itself more often in those with pre-existing anxiety. However, this isn't consistent with everyone, with some users reporting no euphoric qualities at all. It should also be noted that if this effect is experienced, it is typically more subtle in nature in comparison to more potent & faster acting benzodiazepines, such as alprazolam (Xanax.)
Visual effects 
-
- Visual acuity suppression - Like many depressants, diazepam is known to cause blurred or otherwise suppressed visual acuity. This is less likely to occur than other benzodiazepines, however it can still present itself at higher doses, or if the user has a low tolerance.
Paradoxical effects 
-
Paradoxical reactions to benzodiazepines such as increased seizures (in epileptics), aggression, increased anxiety, violent behavior, loss of impulse control, irritability and suicidal behavior sometimes occur (although they are rare in the general population, with an incidence rate below 1%).[23][24]
These paradoxical effects occur with greater frequency in recreational abusers, individuals with mental disorders, children, and patients on high-dosage regimes.[25][26]
Cognitive effects 
-
The general head space of diazepam is described by many as one of intense sedation and decreased inhibition.
The most prominent of these cognitive effects generally include:
- Anxiety suppression
- Disinhibition
- Delusions of sobriety - This is the false belief that one is perfectly sober despite obvious evidence to the contrary such as severe cognitive impairment and an inability to fully communicate with others. It most commonly occurs at heavy dosages.
- Thought deceleration
- Analysis suppression
- Amnesia
- Memory suppression - Diazepam primarily suppresses short-term memory, resulting in forgetfulness, and/or disorganized behaviors.
- Compulsive redosing
- Emotion suppression - Although this compound primarily suppresses anxiety, it also dulls other emotions in a manner which is distinct but less intensive than that of antipsychotics.
- Motivation suppression - Due to diazepam's moderate sedation and lethargy, doing any type of activity that requires moving, or high amounts of effort may be difficult to do on this compound, especially at higher doses.
- Language suppression - Diazepam & most benzodiazepines are known to cause slurred speech and difficulty communicating words in a clear fashion.
After effects 
-
- Rebound anxiety - Rebound anxiety is a commonly observed effect with anxiety relieving substances like benzodiazepines. It typically corresponds to the total duration spent under the substance's influence along with the total amount consumed in a given period, an effect which can easily lend itself to cycles of dependence and addiction.
- Dream potentiation[27] or Dream suppression
- Residual sleepiness - While benzodiazepines can be used as an effective sleep-inducing aid, their effects may persist into the morning afterward, which may lead users to feeling "groggy" or "dull" for up to a few hours.
- Thought deceleration
- Thought disorganization
- Irritability
Experience reports
Anecdotal reports which describe the effects of this compound within our experience index include:
- Experience: A combination of diazepam and alcohol
- Experience:40mg Zolpidem / 20mg Diazepam - Please Don't Do This
- Experience:Diazepam (20/10mg, Oral) - Comfortably Drunk
Additional experience reports can be found here:
Toxicity and harm potential
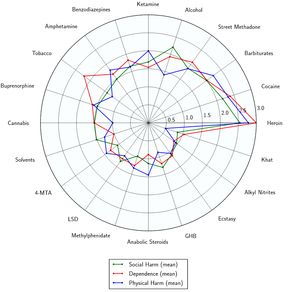
Diazepam has a low toxicity relative to dose.[5] However, it is potentially lethal when mixed with depressants like alcohol or opioids.
It is strongly recommended that one use harm reduction practices, such as volumetric dosing, when using this substance to ensure the administration of the intended dose.
Lethal dosage
The oral LD50 (lethal dose in 50% of the population) of diazepam is 720 mg/kg in mice and 1240 mg/kg in rats.[29] D. J. Greenblatt and colleagues reported in 1978 on two patients who had taken 500 and 2000 mg of diazepam, went into moderately deep comas, and were discharged within 48 hours without having experienced any important complications in spite of having high concentrations of diazepam and its metabolites esmethyldiazepam, oxazepam, and temazepam (according to samples taken in the hospital and as follow-up).[30]
Although not usually fatal when taken alone, a diazepam overdose is considered a medical emergency and generally requires the immediate attention of medical personnel. The antidote for an overdose of diazepam (or any other benzodiazepine) is flumazenil (Anexate). This drug is only used in cases with severe respiratory depression or cardiovascular complications. Because flumazenil is a short-acting drug and the effects of diazepam can last for days, several doses of flumazenil may be necessary. Artificial respiration and stabilization of cardiovascular functions may also be necessary.[31][32][33][11]
Tolerance and addiction potential
Diazepam is extremely physically and psychologically addictive.
Tolerance will develop to the sedative-hypnotic effects within a couple of days of continuous use.[34] After cessation, the tolerance returns to baseline in 7-14 days. However, in certain cases this may take significantly longer in a manner which is proportional to the duration and intensity of one's long-term usage.
Withdrawal symptoms or rebound symptoms may occur after ceasing usage abruptly following a few weeks or longer of steady dosing, and may necessitate a gradual dose reduction.[35][36] For more information on tapering from benzodiazepines in a controlled manner, please see this guide.
Benzodiazepine discontinuation is notoriously difficult; it is potentially life-threatening for individuals using regularly to discontinue use without tapering their dose over a period of weeks. There is an increased risk of hypertension, seizures, and death.[6] Drugs which lower the seizure threshold such as tramadol should be avoided during withdrawal.
Diazepam presents cross-tolerance with all benzodiazepines, meaning that after its consumption all benzodiazepines will have a reduced effect.
Overdose
Benzodiazepine overdose may occur when a benzodiazepine is taken in extremely heavy quantities or concurrently with other depressants. This is particularly dangerous with other GABAergic depressants such as barbiturates and alcohol since they work in a similar fashion, but bind to distinct allosteric sites on the GABAA receptor, thus their effects potentiate one another. Benzodiazepines increase the frequency in which the chlorine ion pore opens on the GABAA receptor while barbiturates increase the duration in which they are open, meaning when both are consumed, the ion pore will open more frequently and stay open longer[37]. Benzodiazepine overdose is a medical emergency that may lead to a coma, permanent brain injury or death if not treated promptly and properly.
Symptoms of a benzodiazepine overdose may include severe thought deceleration, slurred speech, confusion, delusions, respiratory depression, coma or death. Benzodiazepine overdoses may be treated effectively in a hospital environment, with generally favorable outcomes. Benzodiazepine overdoses are sometimes treated with flumazenil, a GABAA antagonist[38], however care is primarily supportive in nature.
Dangerous interactions
Although many drugs are safe on their own, they can become dangerous and even life-threatening when combined with other substances. The list below contains some common potentially dangerous combinations, but may not include all of them. Certain combinations may be safe in low doses of each but still increase the potential risk of death. Independent research should always be done to ensure that a combination of two or more substances is safe before consumption.
- Depressants (1,4-Butanediol, 2-methyl-2-butanol, alcohol, barbiturates, GHB/GBL, methaqualone, opioids) - This combination can result in dangerous or even fatal levels of respiratory depression. These substances potentiate the muscle relaxation, sedation and amnesia caused by one another and can lead to unexpected loss of consciousness at high doses. There is also an increased risk of vomiting during unconsciousness and death from the resulting suffocation. If this occurs, users should attempt to fall asleep in the recovery position or have a friend move them into it.
- Dissociatives - This combination can result in an increased risk of vomiting during unconsciousness and death from the resulting suffocation. If this occurs, users should attempt to fall asleep in the recovery position or have a friend move them into it.
- Stimulants - It is dangerous to combine benzodiazepines with stimulants due to the risk of excessive intoxication. Stimulants decrease the sedative effect of benzodiazepines, which is the main factor most people consider when determining their level of intoxication. Once the stimulant wears off, the effects of benzodiazepines will be significantly increased, leading to intensified disinhibition as well as other effects. If combined, one should strictly limit themselves to only dosing a certain amount of benzodiazepines per hour. This combination can also potentially result in severe dehydration if hydration is not monitored.
Legal status
Internationally, diazepam is a Schedule IV controlled drug under the Convention on Psychotropic Substances.[39] Diazepam is regulated in most countries as a prescription drug.
- Australia: Diazepam is a Schedule 4 substance under the Poisons Standard, making it a prescription only medicine.[40]
- Austria: Diazepam is legal for medical use under the AMG (Arzneimittelgesetz Österreich) and illegal when sold or possessed without a prescription under the SMG (Suchtmittelgesetz Österreich).[citation needed]
- Canada: Diazepam is listed under Schedule IV of the Controlled Drugs and Substances Act[41]
- Czech Republic: Diazepam is a Schedule IV [42] (List 7) substance. Sold exclusively with a prescription "without a blue stripe" (§ 1, g), 1. of Nařízení vlády č. 463/2013 Sb.) [43]
- Germany: Diazepam is controlled under Anlage III BtMG (Narcotics Act, Schedule III) as of August 1, 1986.[44] It can only be prescribed on a narcotic prescription form, except preparations which contain up to 10 mg diazepam in each dosage form and solutions that contain up to 1% and under 250 mg diazepam in total per packaging unit.[45]
- Russia: Diazepam is a Schedule III controlled substance since 2013.[46]
- Switzerland: Diazepam is a controlled substance specifically named under Verzeichnis B. Medicinal use is permitted.[47]
- United Kingdom: Diazepam is classified as a controlled drug and listed under Schedule IV, Part I (CD Benz POM) of the Misuse of Drugs Regulations 2001, allowing possession with a valid prescription. The Misuse of Drugs Act 1971 makes it illegal to possess the substance without a prescription, and, for such purposes, it is classified as a Class C drug.[48]
See also
External links
- Diazepam (Wikipedia)
- Diazepam (Erowid Vault)
- Diazepam (Isomer Design)
- Diazepam (DrugBank)
- Diazepam (Drugs.com)
- Diazepam (Drugs-Forum)
Literature
- Calcaterra, N. E., & Barrow, J. C. (2014). Classics in Chemical Neuroscience: Diazepam (Valium). ACS Chemical Neuroscience, 5(4), 253-260. PMID: 24552479 https://doi.org/10.1021/cn5000056
References
- ↑ Risks of Combining Depressants - TripSit
- ↑ Wick, J. Y. (1 September 2013). "The History of Benzodiazepines". The Consultant Pharmacist. 28 (9): 538–548. doi:10.4140/TCP.n.2013.538. ISSN 0888-5109.
- ↑ Calcaterra, N. E., Barrow, J. C. (16 April 2014). "Classics in Chemical Neuroscience: Diazepam (Valium)". ACS Chemical Neuroscience. 5 (4): 253–260. doi:10.1021/cn5000056. ISSN 1948-7193.
- ↑ WHO Model List (2005) | http://whqlibdoc.who.int/hq/2005/a87017_eng.pdf
- ↑ 5.0 5.1 Mandrioli, R., Mercolini, L., Raggi, M. A. (October 2008). "Benzodiazepine metabolism: an analytical perspective". Current Drug Metabolism. 9 (8): 827–844. doi:10.2174/138920008786049258. ISSN 1389-2002.
- ↑ 6.0 6.1 Lann, M. A., Molina, D. K. (June 2009). "A fatal case of benzodiazepine withdrawal". The American Journal of Forensic Medicine and Pathology. 30 (2): 177–179. doi:10.1097/PAF.0b013e3181875aa0. ISSN 1533-404X.
- ↑ Kahan, M., Wilson, L., Mailis-Gagnon, A., Srivastava, A. (November 2011). "Canadian guideline for safe and effective use of opioids for chronic noncancer pain. Appendix B-6: Benzodiazepine Tapering". Canadian Family Physician. 57 (11): 1269–1276. ISSN 0008-350X.
- ↑ Unseld, E.; Krishna, D. R.; Fischer, C.; Klotz, U. (1989). "Detection of desmethyldiazepam and diazepam in brain of different species and plants". Biochemical Pharmacology. 38 (15): 2473–2478. doi:10.1016/0006-2952(89)90091-9. eISSN 1873-2968. ISSN 0006-2952. OCLC 01536391. PMID 2502983.
- ↑ [1]
- ↑ 10.0 10.1 Sample, I. (2005), Leo Sternbach, retrieved 8 July 2019
- ↑ 11.0 11.1 Barondes, S. H. (2003). Better than Prozac: creating the next generation of psychiatric drugs. Oxford University Press. ISBN 9780195151305.
- ↑ Marshall, K. P., Georgievskava, Z., Georgievsky, I. (June 2009). "Social reactions to Valium and Prozac: A cultural lag perspective of drug diffusion and adoption". Research in Social and Administrative Pharmacy. 5 (2): 94–107. doi:10.1016/j.sapharm.2008.06.005. ISSN 1551-7411.
- ↑ Mariani, M. (2015), Poison Pill - How the American opiate epidemic was started by one pharmaceutical company., retrieved 10 January 2018
- ↑ Mikota SK, Plumb DC (2005). "Diazepam". The Elephant Formulary. Elephant Care International. Archived from the original on 8 September 2005.
- ↑ Taft, W. C., DeLorenzo, R. J. (May 1984). "Micromolar-affinity benzodiazepine receptors regulate voltage-sensitive calcium channels in nerve terminal preparations". Proceedings of the National Academy of Sciences. 81 (10): 3118–3122. doi:10.1073/pnas.81.10.3118. ISSN 0027-8424.
- ↑ "COMMUNICATIONS". British Journal of Pharmacology. 84 (1): 1P–77P. January 1985. doi:10.1111/j.1476-5381.1985.tb17368.x. ISSN 0007-1188.
- ↑ "National Highway Traffic Safety Administration Drugs and Human Performance Fact Sheet- Diazepam". Archived from the original on 27 March 2017.
- ↑ Tan, K. R., Rudolph, U., Lüscher, C. (April 2011). "Hooked on benzodiazepines: GABAA receptor subtypes and addiction". Trends in Neurosciences. 34 (4): 188–197. doi:10.1016/j.tins.2011.01.004. ISSN 0166-2236.
- ↑ Whirl-Carrillo, M., McDonagh, E. M., Hebert, J. M., Gong, L., Sangkuhl, K., Thorn, C. F., Altman, R. B., Klein, T. E. (October 2012). "Pharmacogenomics Knowledge for Personalized Medicine". Clinical Pharmacology & Therapeutics. 92 (4): 414–417. doi:10.1038/clpt.2012.96. ISSN 0009-9236.
- ↑ Zakusov, V. V., Ostrovskaya, R. U., Kozhechkin, S. N., Markovich, V. V., Molodavkin, G. M., Voronina, T. A. (October 1977). "Further evidence for GABA-ergic mechanisms in the action of benzodiazepines". Archives Internationales De Pharmacodynamie Et De Therapie. 229 (2): 313–326. ISSN 0003-9780.
- ↑ Haefely, W. (29 June 1984). "Benzodiazepine interactions with GABA receptors". Neuroscience Letters. 47 (3): 201–206. doi:10.1016/0304-3940(84)90514-7. ISSN 0304-3940.
- ↑ McLean, M. J., Macdonald, R. L. (February 1988). "Benzodiazepines, but not beta carbolines, limit high frequency repetitive firing of action potentials of spinal cord neurons in cell culture". The Journal of Pharmacology and Experimental Therapeutics. 244 (2): 789–795. ISSN 0022-3565.
- ↑ Saïas, T., Gallarda, T. (September 2008). "[Paradoxical aggressive reactions to benzodiazepine use: a review]". L’Encephale. 34 (4): 330–336. doi:10.1016/j.encep.2007.05.005. ISSN 0013-7006.
- ↑ Paton, C. (December 2002). "Benzodiazepines and disinhibition: a review". Psychiatric Bulletin. 26 (12): 460–462. doi:10.1192/pb.26.12.460. ISSN 0955-6036.
- ↑ Bond, A. J. (1 January 1998). "Drug- Induced Behavioural Disinhibition". CNS Drugs. 9 (1): 41–57. doi:10.2165/00023210-199809010-00005. ISSN 1179-1934.
- ↑ Drummer, O. H. (February 2002). "Benzodiazepines - Effects on Human Performance and Behavior". Forensic Science Review. 14 (1–2): 1–14. ISSN 1042-7201.
- ↑ Goyal, S. (14 March 1970). "Drugs and dreams". Canadian Medical Association Journal. 102 (5): 524. ISSN 0008-4409.
- ↑ Nutt, D., King, L. A., Saulsbury, W., Blakemore, C. (24 March 2007). "Development of a rational scale to assess the harm of drugs of potential misuse". The Lancet. 369 (9566): 1047–1053. doi:10.1016/S0140-6736(07)60464-4. ISSN 0140-6736.
- ↑ http://www.drugs.com/diazepam.html
- ↑ Greenblatt, D. J., Woo, E., Allen, M. D., Orsulak, P. J., Shader, R. I. (20 October 1978). "Rapid recovery from massive diazepam overdose". JAMA. 240 (17): 1872–1874. ISSN 0098-7484.
- ↑ Diazepam (PIM 181)
- ↑ http://www.drugs.com/diazepam.html
- ↑ Diazepam (Diazepam Injection): Uses, Dosage, Side Effects, Interactions, Warning
- ↑ Janicak, P. G., Marder, S. R., Pavuluri, M. N. (25 October 2010). Principles and Practice of Psychopharmacotherapy. Lippincott Williams & Wilkins. ISBN 9781605475653.
- ↑ Verster, J. C., Volkerts, E. R. (7 June 2006). "Clinical Pharmacology, Clinical Efficacy, and Behavioral Toxicity of Alprazolam: A Review of the Literature". CNS Drug Reviews. 10 (1): 45–76. doi:10.1111/j.1527-3458.2004.tb00003.x. ISSN 1080-563X.
- ↑ Galanter, M., Kleber, H. D. (2008). The American Psychiatric Publishing Textbook of Substance Abuse Treatment. American Psychiatric Pub. ISBN 9781585622764.
- ↑ Twyman, R. E., Rogers, C. J., Macdonald, R. L. (March 1989). "Differential regulation of gamma-aminobutyric acid receptor channels by diazepam and phenobarbital". Annals of Neurology. 25 (3): 213–220. doi:10.1002/ana.410250302. ISSN 0364-5134.
- ↑ Hoffman, E. J., Warren, E. W. (September 1993). "Flumazenil: a benzodiazepine antagonist". Clinical Pharmacy. 12 (9): 641–656; quiz 699–701. ISSN 0278-2677.
- ↑ International Narcotics Control Board (2003) | http://infoespai.org/wp-content/uploads/2014/12/green.pdf
- ↑ "Poisons Standard December 2019". Office of Parliamentary Counsel. November 14, 2019. Retrieved December 28, 2019.
- ↑ https://laws-lois.justice.gc.ca/eng/acts/C-38.8/page-12.html#h-95661
- ↑ https://eur-lex.europa.eu/resource.html?uri=cellar:6b5e9beb-1d9b-11ea-95ab-01aa75ed71a1.0001.02/DOC_1&format=PDF
- ↑ https://www.zakonyprolidi.cz/cs/2013-463
- ↑ "Zweite Verordnung zur Änderung betäubungsmittelrechtlicher Vorschriften" (PDF). Bundesgesetzblatt Jahrgang 1986 Teil I (in German). Bundesanzeiger Verlag. July 29, 1986. Retrieved December 26, 2019.
- ↑ "Anlage III BtMG" (in German). Bundesministerium der Justiz und für Verbraucherschutz. Retrieved December 26, 2019.
- ↑ Постановление Правительства РФ от 04.02.2013 N 78 “О внесении изменений в некоторые акты Правительства Российской Федерации” - КонсультантПлюс9
- ↑ "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien" (in German). Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved January 1, 2020.
- ↑ Drugs licensing