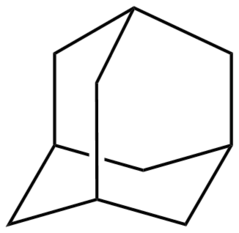
Adamantanes
This article is a stub. As such, it may contain incomplete or wrong information. You can help by expanding it. |
Adamantanes are a chemical class of organic compounds that are based on the adamantane structure. Members of this class have various pharmacological actions which includes dopaminergic stimulation and NMDA receptor antagonism. Memantine and bromantane are two prominent members of this class.
History
In the 1960s, the adamantane derivative amantadine was developed as an antiviral drug for the treatment of influenza. Other adamantane antivirals subsequently followed, such as rimantadine and adapromine.[1] It was serendipitously discovered in 1969 that amantadine possesses central dopaminergic psychostimulant-like properties,[2] and subsequent investigation revealed that rimantadine and adapromine also possess such properties.[3]
Amantadine was then developed and introduced for the treatment of Parkinson's disease due to its ability to increase dopamine levels in the brain. It has also notably since been used to help alleviate fatigue in multiple sclerosis. With the knowledge of the dopaminergic psychostimulant effects of the adamantane derivatives, in the 1980s was developed bromantane as "a drug having psychoactivating and adaptogen properties under complicated conditions".[4]
Adamantane was identified as a key structural subunit in several synthetic cannabinoid designer drugs, namely AB-001 and SDB-001.[5]
Chemistry
This chemistry section is incomplete. You can help by adding to it. |
Adamantane molecules consists of three connected cyclohexane rings arranged in the "armchair" configuration.
Pharmacology
The psychotropic activity spectrum of many adamantane-containing compounds is determined by the nature of their amine substituents, their positions in the polycyctic nucleus, and the basicity of the amino groups. Many have been demonstrated to possesses central dopaminergic psychostimulant-like properties. Some adamantanes produce blocking of the open channels of NMDA and n-cholinergic receptors. It was found that some the derivatives of adamantane markedly increases the production of acetylcholine in the frontal cortex and hippocampus.[3]
In 2004, it was discovered that amantadine and memantine bind to and act as agonists of the σ1 receptor (Ki = 7.44 µM and 2.60 µM, respectively), and that activation of the σ1 receptor is involved in the central dopaminergic effects of amantadine at therapeutically relevant concentrations. These findings may also extend to the other adamantanes such as adapromine, rimantadine, and bromantane, and might potentially explain the psychostimulant-like effects of this family of compounds.[6]
Examples
NMDA receptor antagonists
Stimulants
Depressants
Synthetic cannabinoids
See also
External links
References
- ↑ Mandell, L., Woodhead, M., Ewig, S., Torres, A. (27 October 2006). Respiratory Infections. CRC Press. ISBN 9780340816943.
- ↑ Brandt, T., Caplan, L. R., Dichgans, J., Diener, H.-C., Kennard, C. (3 January 2003). Neurological Disorders: Course and Treatment. Gulf Professional Publishing. ISBN 9780121258313.
- ↑ 3.0 3.1 Spasov, A. A., Khamidova, T. V., Bugaeva, L. I., Morozov, I. S. (1 January 2000). "Adamantane derivatives: Pharmacological and toxicological properties (review)". Pharmaceutical Chemistry Journal. 34 (1): 1–7. doi:10.1007/BF02524549. ISSN 1573-9031.
- ↑ Oliynyk, S., Oh, S. (September 2012). "The Pharmacology of Actoprotectors: Practical Application for Improvement of Mental and Physical Performance". Biomolecules & Therapeutics. 20 (5): 446–456. doi:10.4062/biomolther.2012.20.5.446. ISSN 1976-9148.
- ↑ Banister, S. D., Wilkinson, S. M., Longworth, M., Stuart, J., Apetz, N., English, K., Brooker, L., Goebel, C., Hibbs, D. E., Glass, M., Connor, M., McGregor, I. S., Kassiou, M. (17 July 2013). "The synthesis and pharmacological evaluation of adamantane-derived indoles: cannabimimetic drugs of abuse". ACS chemical neuroscience. 4 (7): 1081–1092. doi:10.1021/cn400035r. ISSN 1948-7193.
- ↑ Peeters, M., Romieu, P., Maurice, T., Su, T.-P., Maloteaux, J.-M., Hermans, E. (April 2004). "Involvement of the sigma 1 receptor in the modulation of dopaminergic transmission by amantadine". The European Journal of Neuroscience. 19 (8): 2212–2220. doi:10.1111/j.0953-816X.2004.03297.x. ISSN 0953-816X.